
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
9th Edition
ISBN: 9781292151229
Author: Wade, LeRoy G.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.37SP
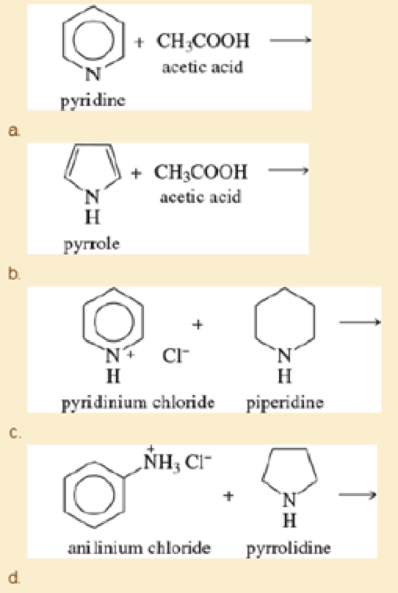
Complete the following proposed acid-base reactions, and predict whether the reactants or products are favored.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Rank the following compounds in order of increasing acidity (1 = least acidic, 3 = most acidic) and in the space provided use resonance (of the conjugate base) to explain why the compound you have labelled “3” is the most acidic.
Use the information below to identify and explain the relationship between the basicity of the two nucleophiles and their nucleophilicities
Show how acid derivatives hydrolyze to carboxylic acids under either acidic or basicconditions. Explain why some acid derivatives (amides, for example) require muchstronger conditions for hydrolysis than other derivatives.
Chapter 19 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
Ch. 19.2A - Prob. 19.1PCh. 19.2B - Prob. 19.2PCh. 19.2B - Give correct names for the following amines:Ch. 19.3 - Prob. 19.4PCh. 19.4 - Prob. 19.5PCh. 19.6 - Rank each set of compounds in order of increasing...Ch. 19.8A - Prob. 19.7PCh. 19.8C - Prob. 19.8PCh. 19.8C - Prob. 19.9PCh. 19.8D - a. Show how fragmentation occurs to give the base...
Ch. 19.10B - Propose a mechanism for nitration of pyridine at...Ch. 19.10B - Prob. 19.12PCh. 19.10C - Prob. 19.13PCh. 19.10C - Prob. 19.14PCh. 19.11 - Propose a mechanism to show the individual...Ch. 19.11 - Prob. 19.16PCh. 19.12 - Give the products expected from the following...Ch. 19.13 - Prob. 19.18PCh. 19.13 - Prob. 19.19PCh. 19.14 - Prob. 19.20PCh. 19.15 - Prob. 19.21PCh. 19.15 - Prob. 19.22PCh. 19.16 - Prob. 19.23PCh. 19.17 - Prob. 19.24PCh. 19.17 - Prob. 19.25PCh. 19.18 - Prob. 19.26PCh. 19.19 - Prob. 19.27PCh. 19.20A - Addition of one equivalent of ammonia to...Ch. 19.20A - Prob. 19.29PCh. 19.20B - Show how you would accomplish the following...Ch. 19.20C - Prob. 19.31PCh. 19 - For each compound, 1. classify the...Ch. 19 - Prob. 19.33SPCh. 19 - Within each structure, rank the indicated...Ch. 19 - In each pair of compounds, select the stronger...Ch. 19 - Which of the following compounds are capable of...Ch. 19 - Complete the following proposed acid-base...Ch. 19 - Predict the products of the following reactions:...Ch. 19 - Prob. 19.39SPCh. 19 - Show how m-toluidine can be converted to the...Ch. 19 - The mass spectrum of tert-butylamine follows shows...Ch. 19 - Prob. 19.42SPCh. 19 - The following drugs are synthesized using the...Ch. 19 - Prob. 19.44SPCh. 19 - Synthesize from benzene. (Hint: All of these...Ch. 19 - Propose mechanisms for the following reactions.Ch. 19 - Prob. 19.47SPCh. 19 - Prob. 19.48SPCh. 19 - Prob. 19.49SPCh. 19 - Show how you can synthesize the following...Ch. 19 - Prob. 19.51SPCh. 19 - The alkaloid coniine has been isolated from...Ch. 19 - A chemist is summoned to an abandoned...Ch. 19 - Pyrrole undergoes electrophilic aromatic...Ch. 19 - Prob. 19.55SPCh. 19 - Prob. 19.56SPCh. 19 - An unknown compound shows a weak molecular ion at...Ch. 19 - A compound of formula C11H16N2 gives the IR,...Ch. 19 - (A true story.) A drug user responded to an ad...Ch. 19 - Prob. 19.60SPCh. 19 - Prob. 19.61SPCh. 19 - Prob. 19.62SPCh. 19 - Prob. 19.63SPCh. 19 - Prob. 19.64SPCh. 19 - Prob. 19.65SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Arrange these carboxylate ions in order of decreasing basicity.arrow_forwardRank the following in order of decreasing basicity and explain why (based on their structures). hydroxide ion, ethoxide ion, phenoxide ion, and carboxylate ionarrow_forwardAnswer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D below each structure.arrow_forward
- Arrange the following molecules in increasing order of acidity. Base it only on their structural differences and explain how it is so. 1. HF, CH3CH2CH2OH, CH3CH2COOH 2. Ethyl amine, Ethanol, Propanearrow_forwardPlease complete the following question. There is no need to provide an explaination. 15arrow_forwardPredict the stronger acid between the pair of acids and explain your answer. a.I3CCH2CH2COOH and CH3CH2CCL2COOH.arrow_forward
- From 1 being the least acidic and 4 being the most acidic, arrange the following organic compounds in terms of increasing acidityarrow_forwardWhich is true regarding the direction of the following reaction? CH3COOH (aq) + H2PO-4 <<>>> CH3COO- + H3PO4 a) the reaction favors the reactant side b) the reaction favors the product side c) the reaction favors both reactants and products equally d) the table of acidity does not proviede enough information to answer this questionarrow_forwardRegarding to the following compounds: NH3, H2O, H2SA) Rank the following compounds in order of increasing acidityB) Mention their conjugate bases and rank according to increasing their basicity orderarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY