
Interpretation:
The structures for cis -3-hexene and trans -3-hexene should be drawn.
Concept introduction:
The molecule with same molecular formula and connectivity but differ in spatial arrangements of atoms in the molecule are said to be geometric isomers of each other.
Answer to Problem 27SSC
The structures for cis -3-hexene and trans -3-hexene is:
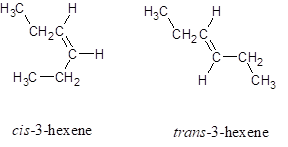
Explanation of Solution
Geometric isomerism, or cis-trans, isomerism is most common in
In cis- isomer, the two identical groups or atoms are close to each other whereas in trans- isomer two identical groups or atoms are farther apart.
The structures for cis -3-hexene and trans -3-hexene (hex represents the parent chain contains six carbon atoms and 3 represents the position of double bond in the chain) is:
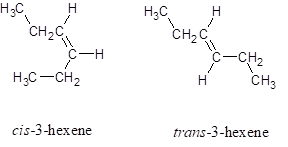
Chapter 21 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Inorganic Chemistry
General Chemistry: Principles and Modern Applications (11th Edition)
Organic Chemistry (8th Edition)
Chemistry: Structure and Properties
Organic Chemistry (9th Edition)
Chemistry: Structure and Properties (2nd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





