
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 83E
Give the empirical formula for each of the compounds represented below.
a. 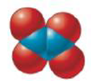
b. 
c. 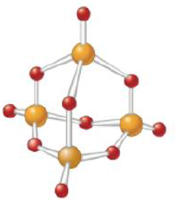
d. 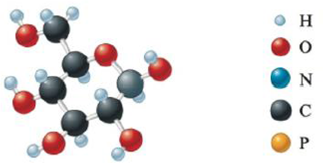
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 3 Solutions
Chemistry
Ch. 3 - Prob. 1RQCh. 3 - Atomic masses are relative masses. What does this...Ch. 3 - The atomic mass of boron (B) is given in the...Ch. 3 - What three conversion factors and in what order...Ch. 3 - Fig. 5-5 illustrates a schematic diagram of a...Ch. 3 - What is the difference between the empirical and...Ch. 3 - Consider the hypothetical reaction between A2 and...Ch. 3 - Prob. 8RQCh. 3 - Consider the following mixture of SO2(g) and...Ch. 3 - Why is the actual yield of a reaction often less...
Ch. 3 - The following are actual student responses to the...Ch. 3 - What information do we get from a chemical...Ch. 3 - True or false? The atom with the largest subscript...Ch. 3 - You have a 20.0-g sample of silver metal. You are...Ch. 3 - You are making cookies and are missing a key...Ch. 3 - Nitrogen gas (N2) and hydrogen gas (H2) react to...Ch. 3 - For the preceding question, which of the following...Ch. 3 - You know that chemical A reacts with chemical B....Ch. 3 - Prob. 9ALQCh. 3 - Consider an iron bar on a balance as shown. As the...Ch. 3 - You may have noticed that water sometimes drips...Ch. 3 - Prob. 12ALQCh. 3 - What is true about the chemical properties of the...Ch. 3 - Is there a difference between a homogeneous...Ch. 3 - Chlorine exists mainly as two isotopes, 37Cl and...Ch. 3 - The average mass of a carbon atom is 12.011....Ch. 3 - Can the subscripts in a chemical formula be...Ch. 3 - Consider the equation 2A + B . A2B. If you mix 1.0...Ch. 3 - According to the law of conservation of mass, mass...Ch. 3 - Which of the following pairs of compounds have the...Ch. 3 - Atoms of three different elements are represented...Ch. 3 - In chemistry, what is meant by the term mole? What...Ch. 3 - Which (if any) of the following is (are) true...Ch. 3 - Consider the equation 3A + B C + D. You react 4...Ch. 3 - The atomic masses in die periodic table are...Ch. 3 - Avogadros number, molar mass, and the chemical...Ch. 3 - If you had a mole of U.S. dollar bills and equally...Ch. 3 - Prob. 28QCh. 3 - Which of the following compounds have the same...Ch. 3 - What is the difference between the molar mass and...Ch. 3 - How is the mass percent of elements in a compound...Ch. 3 - A balanced chemical equation contains a large...Ch. 3 - Prob. 33QCh. 3 - Hydrogen gas and oxygen gas react to form water,...Ch. 3 - What is the theoretical yield for a reaction, and...Ch. 3 - What does it mean to say a reactant is present in...Ch. 3 - Consider the following generic reaction: A2B2 + 2C...Ch. 3 - Consider the following generic reaction:...Ch. 3 - An element consists of 1.40% of an isotope with...Ch. 3 - An element X bas five major isotopes, which are...Ch. 3 - The element rhenium (Re) bas two naturally...Ch. 3 - Assume silicon has three major isotopes in nature...Ch. 3 - The element europium exists in nature as two...Ch. 3 - The element silver (Ag) has two naturally...Ch. 3 - The mass spectrum of bromine (Br2) consists of...Ch. 3 - The stable isotopes of iron arc 54Fe, 56Fe, 57Fe,...Ch. 3 - Calculate the mass of 500. atoms of iron (Fe).Ch. 3 - What number of Fe atoms and what amount (moles) of...Ch. 3 - Diamond is a natural form of pure carbon. What...Ch. 3 - Chromium (Cr) is a metal that is added to steel to...Ch. 3 - Selective serotonin reuptake inhibitors (SSRIs)...Ch. 3 - The Freons are a class of compounds containing...Ch. 3 - Calculate the molar mass of the following...Ch. 3 - Calculate the molar mass of the following...Ch. 3 - What amount (moles) of compound is present in 1.00...Ch. 3 - What amount (moles) of compound is present in 1.00...Ch. 3 - What mass of compound is present in 5.00 moles of...Ch. 3 - What mass of compound is present in 5.00 moles of...Ch. 3 - Prob. 59ECh. 3 - Prob. 60ECh. 3 - What number of molecules (or formula units) are...Ch. 3 - What number of molecules (or formula units) are...Ch. 3 - What number of atoms of nitrogen are present in...Ch. 3 - Prob. 64ECh. 3 - Freon- 12 (CCI2F2) is used as a refrigerant in air...Ch. 3 - Prevacid is used to treat gastroesophageal reflux...Ch. 3 - What amount (moles) is represented by each of...Ch. 3 - What number of atoms of nitrogen are present in...Ch. 3 - Complete the following table.Ch. 3 - Ascorbic acid, or vitamin C (C6H8O6), is an...Ch. 3 - The molecular formula of acetylsalicylic acid...Ch. 3 - Chloral hydrate (C2H3Cl3O2) is a drug formerly...Ch. 3 - Dimethylnitrosamine, (CH3)2N2O , is a carcinogenic...Ch. 3 - Calculate the percent composition by mass of the...Ch. 3 - In 1987 the first substance to act as a...Ch. 3 - The percent by mass of nitrogen for a compound is...Ch. 3 - Arrange the following substances in order of...Ch. 3 - Fungal laccase, a blue protein found in...Ch. 3 - Hemoglobin is the protein that transports oxygen...Ch. 3 - Express the composition of each of the following...Ch. 3 - Considering your answer to Exercise 79, which type...Ch. 3 - Give the empirical formula for each of the...Ch. 3 - Determine the molecular formulas to which the...Ch. 3 - A compound that contains only carbon, hydrogen,...Ch. 3 - The most common form of nylon (nylon-6) is 63.68%...Ch. 3 - There are two binary compounds of mercury and...Ch. 3 - A sample of urea contains 1.121 g N, 0.161 g H,...Ch. 3 - A compound containing only sulfur and nitrogen is...Ch. 3 - Determine the molecular formula of a compound that...Ch. 3 - A compound contains 47.08% carbon, 6.59% hydrogen,...Ch. 3 - Maleic acid is an organic compound composed of...Ch. 3 - One of the components that make up common table...Ch. 3 - A compound contains only C, H, and N. Combustion...Ch. 3 - Cumene is a compound containing only carbon and...Ch. 3 - A compound contains only carbon, hydrogen, and...Ch. 3 - Give the balanced equation for each of the...Ch. 3 - Give the balanced equation for each of the...Ch. 3 - A common demonstration in chemistry courses...Ch. 3 - Iron oxide ores, commonly a mixture of FeO and...Ch. 3 - Balance the following equations: a. Ca(OH)2(aq) +...Ch. 3 - Balance each of the following chemical equations....Ch. 3 - Balance the following equations representing...Ch. 3 - Balance the following equations: a. Cr(s) + S8(s) ...Ch. 3 - Silicon is produced for the chemical and...Ch. 3 - Glass is a mixture of several compounds, but a...Ch. 3 - Phosphorus occurs naturally in the form of...Ch. 3 - The sugar sucrose, which is present in many fruits...Ch. 3 - Over the years, the thermite reaction has been...Ch. 3 - The reaction between potassium chlorate and red...Ch. 3 - The reusable booster rockets of the U.S. space...Ch. 3 - One of relatively few reactions that takes place...Ch. 3 - Prob. 113ECh. 3 - a. Write die balanced equation for the combustion...Ch. 3 - Elixirs such as Atka-Seltzer use the reaction of...Ch. 3 - Aspirin (C9H8O4) is synthesized by reacting...Ch. 3 - Bacterial digestion is an economical method of...Ch. 3 - Phosphorus can be prepared from calcium phosphate...Ch. 3 - Coke is an impure form of carbon that is often...Ch. 3 - The space shuttle environmental control system...Ch. 3 - Consider the reaction between NO(g) and O2(g)...Ch. 3 - Consider the following reaction:...Ch. 3 - Ammonia is produced from the reaction of nitrogen...Ch. 3 - Consider the following unbalanced equation:...Ch. 3 - Hydrogen peroxide is used as a cleansing agent in...Ch. 3 - Silver sulfadiazine bum-treating cream creates a...Ch. 3 - Hydrogen cyanide is produced industrially from the...Ch. 3 - Acrylonitrile C3H3N) is the starting material for...Ch. 3 - Aluminum reacts with bromine, producing aluminum...Ch. 3 - Hexamethylenediamine (C6H16N2) is one of the...Ch. 3 - The reaction of ethane gas (C2H6) with chlorine...Ch. 3 - DDT, an insecticide harmful to fish, birds, and...Ch. 3 - Bornite (Cu3FeS3) is a copper ore used in the...Ch. 3 - Prob. 134ECh. 3 - In using a mass spectrometer, a chemist sees a...Ch. 3 - Boron consists of two isotopes, 10B and 11B....Ch. 3 - A given sample of a xenon fluoride compound...Ch. 3 - Aspartame is an artificial sweetener that is 160...Ch. 3 - Anabolic steroids are performance enhancement...Ch. 3 - In the spring of 1984, concern arose over the...Ch. 3 - You find a compound composed only of element X and...Ch. 3 - What mass of sodium hydroxide has the same number...Ch. 3 - The compound adrenaline contains 56.79% C, 6.56%...Ch. 3 - Adipic acid is an organic compound composed of...Ch. 3 - Vitamin B12, cyanocobalamin, is essential for...Ch. 3 - Some bismuth tablets, a medication used to treat...Ch. 3 - The empirical formula of styrene is CH; the molar...Ch. 3 - Terephthalic acid is an important chemical used in...Ch. 3 - A sample of a hydrocarbon (a compound consisting...Ch. 3 - A binary compound between an unknown element E and...Ch. 3 - A 0.755-g sample of hydrated copper(II) sulfate...Ch. 3 - ABS plastic is a tough, hard plastic used in...Ch. 3 - A sample of LSD (D-lysergic acid diethylamide,...Ch. 3 - Methane (CH4) is the main component of marsh gas....Ch. 3 - A potential fuel for rockets is a combination of...Ch. 3 - A 0.4230-g sample of impure sodium nitrate was...Ch. 3 - Prob. 157AECh. 3 - Commercial brass, an alloy of Zn and Cu, reacts...Ch. 3 - Vitamin A has a molar mass of 286.4 g/mol and a...Ch. 3 - You have seven closed containers, each with equal...Ch. 3 - A substance X2Z has the composition (by mass) of...Ch. 3 - Consider samples of phosphine (PH3), water (H2O),...Ch. 3 - Calculate the number of moles for each compound in...Ch. 3 - Arrange the following substances in order of...Ch. 3 - Para-cresol, a substance used as a disinfectant...Ch. 3 - A compound with molar mass 180.1 g/mol has the...Ch. 3 - Prob. 167CWPCh. 3 - Consider the following unbalanced chemical...Ch. 3 - Sulfur dioxide gas reacts with sodium hydroxide to...Ch. 3 - Gallium arsenide, GaAs, has gained widespread use...Ch. 3 - Consider the following data for three binary...Ch. 3 - Natural rubidium has the average mass of 85.4678 u...Ch. 3 - A compound contains only carbon, hydrogen,...Ch. 3 - Nitric acid is produced commercially by the...Ch. 3 - When the supply of oxygen is limited, iron metal...Ch. 3 - A 9.780-g gaseous mixture contains ethane (C2H6)...Ch. 3 - Zinc and magnesium metal each reacts with...Ch. 3 - A gas contains a mixture of NH3(g) and N2H4(g),...Ch. 3 - Consider a gaseous binary compound with a molar...Ch. 3 - A 2.25-g sample of scandium metal is reacted with...Ch. 3 - Prob. 181CPCh. 3 - The aspirin substitute, acetaminophen (C8H9O2N),...Ch. 3 - An element X forms both a dichloride (XCl2) and a...Ch. 3 - When M2S3(s) is heated in air, it is converted to...Ch. 3 - When aluminum metal is heated with an element from...Ch. 3 - Consider a mixture of potassium chloride and...Ch. 3 - Lanthanum was reacted with hydrogen in a given...Ch. 3 - A 1.500-g sample of a mixture containing only Cu2O...Ch. 3 - Ammonia reacts with O2 to form either NO(g) or...Ch. 3 - You take 1.00 g of an aspirin tablet (a compound...Ch. 3 - With the advent of techniques such as scanning...Ch. 3 - Tetrodotoxin is a toxic chemical found in fugu...Ch. 3 - An iortic compound MX3 is prepared according to...Ch. 3 - The compound As2I4 is synthesized by reaction of...Ch. 3 - A 2.077-g sample of an element, which has an...Ch. 3 - Consider the following balanced chemical equation:...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- . How many atoms of carbon are present in 1 .0 g of CH4O ? How many atoms of carbon are present in 1.0 of CH3CH2OH ? How many atoms of nitrogen are present in 25.0 g of CO( NH2)2 ?arrow_forwarda How many molecules are in 3.61g F2? b How many atoms are in 3.61g F2? c How many atoms are in 3.61g F? d What is the mass of 3.611023 F atoms? e What is the mass of 3.611023 F2 molecules?arrow_forwardConsidering your answer to Exercise 79, which type of formula, empirical or molecular, can be obtained from elemental analysis that gives percent composition?arrow_forward
- f you have equal mole samples of NO2 and F2 , which of the following must be true? l type='a'> The number of molecules in each sample is the same. i>The number of atoms in each sample is the same. i>The masses of the samples are the same. i>6.0221023 molecules are present in each sample.arrow_forwardDetermine a chemical formula from elemental analysis (i.e., from % composition).arrow_forwardSilver is often extracted from ores such as K[Ag(CN)2] and then recovered by the reaction 2K[Ag(CN)2](aq)+Zn(s)2Ag(s)+Zn(CN)2(aq)+2KCN(aq) (a) How many molecules of Zn(CN)2 are produced by the reaction of 35.27 g of K[Ag(CN)2]? (b) What mass of Zn(CN)2 is produced?arrow_forward
- How many molecules of C2H4Cl2 can be prepared from 15C2H4 molecules and 8Cl2 molecules?arrow_forwardUse the average atomic masses given inside the front cover of this book to calculate the mass in grains of each of the following samples. l type='a'> 0.251 mole of lithium 1.51 moles of aluminum 8.75102 moles of lead 125 moles of chromium 4.25103 moles of iron 0.000105 mole of magnesiumarrow_forwardAnalysis of a 10.0-g sample of apatite (a major component of tooth enamel) showed that it was made up of 3.99 g Ca, 1.85 g P, 4.14 g O, and 0.020 g H. List these elements based on relative amounts (moles), from smallest to largest.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY