
Concept explainers
Interpretation:The most acidic hydrogen in pair of molecules given below should be circled. Also, molecule that haslower value of
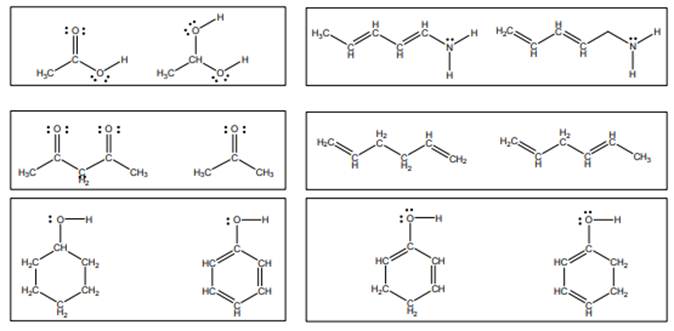
Concept introduction:Lewis structure is representation of molecule in which dots are shown to represent unshared electrons and lines are shown to represent bonds. These lines and dots represent distribution of the electrons in the molecule.
When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Rules to form resonance structure are as follows:
1. Use arrow types 1 and 2 for resonance structure of anions in movement of negative charge.
2. Use only arrow type 3 to move a positive charge for resonance structure of cations.
3. The sigma bond should not be broken. Any atom must not move from its place and total number of electrons must be same in all resonance structures.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- For each pair of compounds, circle the one that is more acidic. Briefly explain why your choice is the more acidic one. Your answer should involve qualitative principles (not just the pKa's given), and you should include structural drawings.arrow_forwardIn the picture, Each of the following compounds has more than one kind of (a-H's). First draw all a-H in, and circle the ones that are more acidic (that is, the ones more likely removed with a suitable base) in each compound. Thank you!arrow_forwardFor the following molecules, identify the acidic hydrogens from among those that are bolded. For each acidic hydrogen that you identify, draw the conjugate base that results from a base removing that acidic hydrogen. For molecules with more than one acidic hydrogen, draw a separate conjugate base for each one.arrow_forward
- For the following groups of molecules (labeled A-C), rank the acidity of the molecules in order from least acidic (3) to most acidic (1). Explain your reasoning.arrow_forwardIn each compound below, rank the labeled protons in order of increasing acidity.arrow_forwardOn which carbon would you find the most acidic proton? The correct option is B but please include a detailed explaination as to why it is B. Thanks:)arrow_forward
- I need help on C D). Consider the three compounds above. Which compound is the most basic? Which compound is least basic? Briefly explain your reasoning on both parts.arrow_forwardRank the compounds below with the strongest acid at the top.arrow_forwardFor each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forward
- Molecule A’s most acidic hydrogen has a pka of 4.1. Point out the most acidic hydrogen and draw it’s conjugate basearrow_forwardI know that the 3rd one (second on the right side) has the highest pka because it's an alcohol and an alcohol is less acidic than a phenol. but I don't understand which compound has the lowest pka (strongest acid)?arrow_forwardEven though it is usually difficult to remove a proton from a neutral nitrogen atom, the proton on this snowman-shaped molecule on the nitrogen adjacent to the carbonyl is acidic! Draw the structure of the conjugate base, the TWO additional resonance contributors, and the resonance hybrid in the indicated boxes.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
