
Concept explainers
(a)
Interpretation: The reasonable mechanism for product B in reaction below should be drawn.
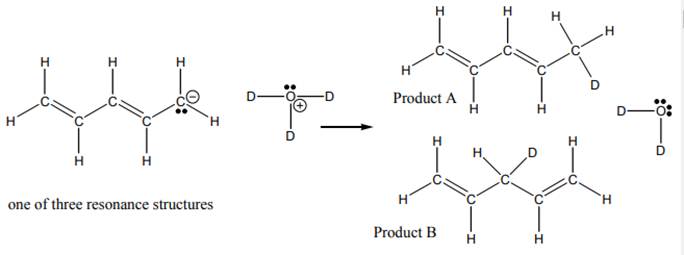
Concept introduction:When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Rules to form resonance structure are as follows:
1. Use arrow types 1 and 2 for resonance structure of anions in movement of negative charge. Arrow type 1 shows the movement of lone pair toward adjacent atom and then converted into
2. Use only arrow type 3 to move a positive charge for resonance structure of cations. Arrow type 3 is used to move
3. The sigma bond should not be broken. Any atom must not move from its place and total number of electrons must be same in all resonance structures.
(b)
Interpretation: The ratio
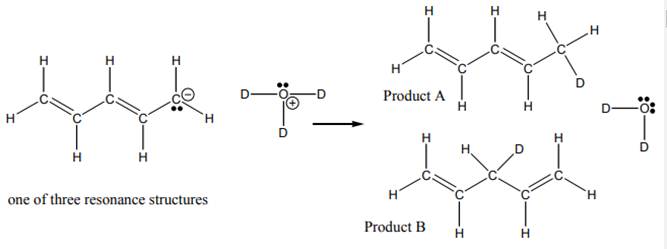
Concept introduction:When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Rules to form resonance structure are as follows:
1. Use arrow types 1 and 2 for resonance structure of anions in movement of negative charge. Arrow type 1 shows the movement of lone pair toward adjacent atom and then converted into
2. Use only arrow type 3 to move a positive charge for resonance structure of cations. Arrow type 3 is used to move
3. The sigma bond should not be broken. Any atom must not move from its place and total number of electrons must be same in all resonance structures.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- While selenic acid has the formula H2SeO4 and thus is directly related to sulfuric acid, telluric acid is best visualized as H6TeO6 or Te(OH)6? a. What is the oxidation state of tellurium in Te(OH)6? b. Despite its structural differences with sulfuric and selenic acid, telluric acid is a diprotic acid with pKa1=7.68 and pKa2=11.29. Telluric add can be prepared by hydrolysis of tellurium hexafluoride according to the equation TeF6(g)+6H2O(l)Te(OH)6(aq)+6HF(aq) Tellurium hexafluoride can be prepared by the reaction of elemental tellurium with fluorine gas: Te(s)+3F2(g)TeF6(g) If a cubic block of tellurium (density = 6.240 g/cm3) measuring 0.545 cm on edge is allowed to react with 2.34 L fluorine gas at 1.06 atm and 25C, what is the pH of a solution of Te(OH)6 formed by dissolving the isolated TeF6(g) in 115 mL solution? Assume 100% yield in all reactions.arrow_forwardWhat mass of CaH2 is necessary to react with water to provide enough hydrogen gas to fill a balloon at 20 C and 0.8 arm pressure with a volume of 4.5 L? The balanced equation is: CaH2(s)+2H2O(l)Ca(OH)2(aq)+2H2(g)arrow_forwardWhy are the lanthanoid elements not found in nature in their elemental forms?arrow_forward
- Give examples of two basic oxides. Write equations illustrating the formation of each oxide from its component elements. Write another chemical equation that illustrates the basic character of each oxide.arrow_forwardMany compounds of Group 3A(13) elements have chemical behavior that reflects an electron deficiency.(a) What is the meaning of electron deficiency?(b) Give two reactions that illustrate this behavior.arrow_forwardHow do Groups 1A(1) and 2A(2) compare with respect to reaction of the metals with water?arrow_forward
- How do atomic properties account for the low densities ofthe Group 1A(1) elements?arrow_forwardWhat is the main characteristic that determines whether or not an element is a main-group element?arrow_forwardIndicate whether each of the following statements is trueor false (a) H2(g) and D2(g) are allotropic forms of hydrogen.(b) ClF3 is an interhalogen compound. (c) MgO(s) isan acidic anhydride. (d) SO2(g) is an acidic anhydride.(e) 2 H3PO4(l)----> H4P2O7(l) + H2O(g) is an example ofa condensation reaction. (f) Tritium is an isotope of theelement hydrogen. (g) 2SO2(g) + O2(g)---> 2SO3(g) is anexample of a disproportionation reaction.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





