
Concept explainers
Interpretation: Any one structure from top row is enough to describe the molecule in top row but all structure is needed from bottom row to describe the molecule in bottom row should be explained.
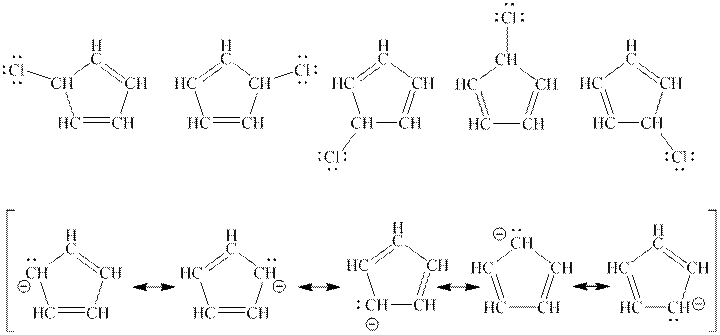
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Rules to form resonance structure are as follows:
1. Use arrow types 1 and 2 for resonance structure of anions in movement of negative charge.
2. Use only arrow type 3 to move a positive charge for resonance structure of cations.
3. The sigma bond should not be broken. Any atom must not move from its place and total number of electrons must be same in all resonance structures.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- Is this structure aromatic, antiaromatic, or nonaromatic? Provide its resonance structure as well.arrow_forwardAdd curved arrows to show how the first resonance structure can beconverted to the second.arrow_forwardHow do I calc the formal chargers of nitrene, oxonium ion, carbene, acetylidearrow_forward
- 1. What is resonance contributor in which one or more atoms bears a formal change and the most stable resonance form? 2.what is the process of distributing electron pairs in a molecule? 3. Are organic molecules which are less sterically hindered more attractive than those which are more sterically hindered?arrow_forwardFollow the curved arrows to draw a second resonance structure for eachspecies.arrow_forwardCalculate the formal charge for each atom that is not carbon or hydrogen in the following molecules.arrow_forward
- Determine the total number of resonance structures. Include the initial structure as one structure in the total.arrow_forwardFollow the curved arrows to draw a second resonance structure for each species.arrow_forwardIn order to determine high and low electron density, is it necessary to include minor resonance structures? The textbook answer says 'significant resonance contributors suggest the areas of highest and lowest electron density.' Does this mean minor structures are also significant?arrow_forward
- I got this marked wrong on an assignment, but I'm not sure why. Please help me find the correct resonance structure.arrow_forwardWhich of the following species (B, C,D) is a valid resonance of A? Use curved arrows to show how A is converted to any valid resonance structure.arrow_forwarddraw all resonances are possible for this structure?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
