
Concept explainers
Interpretation: The Lewis structure of below
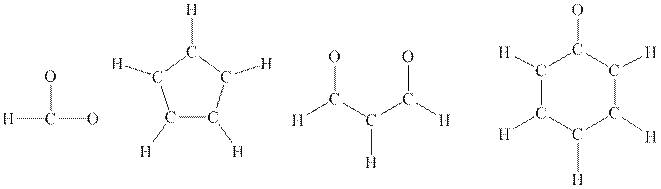
Concept introduction: Lewis structure is representation of molecule in which dots are shown to represent unshared electrons and lines are shown to represent bonds. These lines and dots represent distribution of electrons in the molecule.
When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Rules to form resonance structure are as follows:
1. Use arrow types 1 and 2 for resonance structure of anions in movement of negative charge.
2. Use only arrow type 3 to move a positive charge for resonance structure of cations.
3. The sigma bond should not be broken. Any atom must not move from its place and total number of electrons must be same in all resonance structures.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- Why is this structure wrong and how can it be corrected? It should be neutral and the formal charge for each atom should be 0.arrow_forwardDraw the Lewis structure of the missing product. Make sure to include lone pairs and non-zero formal charges.arrow_forwardFor each of the following structures,1. Draw a Lewis structure; fill in any nonbonding electrons.2. Calculate the formal charge on each atom other than hydrogen.(a) CH3NO(nitromethane)(b) (CH3)3NO(trimethylamine oxide)(c) [N3]-(azide ion)(d) [(CH3)3O]+ (e) CH3NC (f) (CH3)4NBrarrow_forward
- These structures contain at least one error. Identify the errors and suggest a correct structure. All structures should be neutral (uncharged) and formal charge for each atom should be 0.arrow_forwardDraw complete Lewis structures for the following condensed structural formulas.(a) CH3(CH2)3CH(CH3)2 (b) (CH3)2CHCH2Cl(c) CH3CH2COCN (d) CH2CHCHO(e) (CH3)3CCOCHCH2 (f) CH3COCOOHarrow_forwardDraw acceptable Lewis structures, including resonance contributors where appropriate with all non-bonding electrons and formal charges. N2O4 [HCO3]- [F2BrO]+ [H2PO3S]-arrow_forward
- Label each non-hydrogen atom in the structure below with its formal chargearrow_forwardWhat would the resonance structure of these 2 compounds be? And which would be the major?arrow_forwardWhat is the formal charge of each atom (from left to right), neglecting the hydrogens, in the following resonance structure of CH2N2? Select one: a. 0, +1, −1 b. 0, −1, +1 c. −1, −1, +1 d. −1, +1, 0 e. 0, 0, 0arrow_forward
- Write the resonance structure of the following anions. need all 4 parts or else I will downvote soonarrow_forwardWhat are fuctional groups, are they only attached to carbon molecules or molecules that don't contain carbon?arrow_forwardcan someone explain how Molecule B has a tertiary carbon ? Im having troubl with secondary and tertiary i tried to calculate the formal charges thinking that may have something to do with it but I ended up getting the wrong formal charge for molecule B, so now I'm confused.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


