
Concept explainers
(a)
Interpretation:
The hybridization of each carbon atom in following structure should be determined.
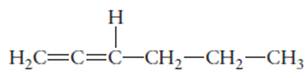
Concept Introduction:
The process of the mixing of orbitals having similar symmetry and energy to results equal number of orbitals is known as hybridization.
The formed orbitals are known as hybrid orbitals.
Carbon belongs to group 4 of the periodic table and its
The electronic configuration of carbon is:
In the hybridization, carbon uses 2s and 2p orbitals.
For example:
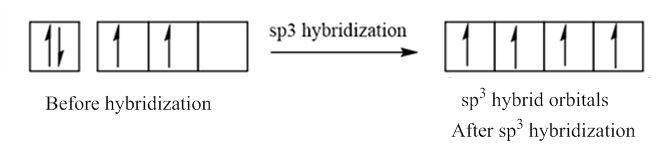
In case of sp3 hybridization, one 2s and three 2p orbitals of carbon combine to form four sp3 hybrid orbitals.
The electronic configuration of carbon before and after sp3 hybridization is:
(b)
Interpretation:
The hybridization of each carbon atom in following structure should be determined.
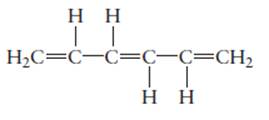
Concept Introduction:
The process of the mixing of orbitals having similar symmetry and energy to results equal number of orbitals is known as hybridization.
The formed orbitals are known as hybrid orbitals.
Carbon belongs to group 4 of the periodic table and its atomic number is 6.
The electronic configuration of carbon is:
In the hybridization, carbon uses 2s and 2p orbitals.
For example:
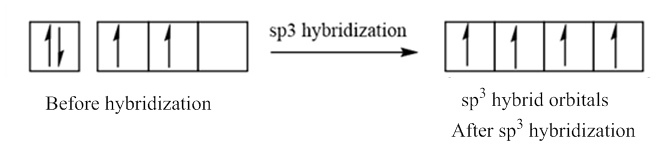
In case of sp3 hybridization, one 2s and three 2p orbitals of carbon combine to form four sp3 hybrid orbitals.
The electronic configuration of carbon before and after sp3 hybridization is:
(c)
Interpretation:
The hybridization of each carbon atom in following structure should be determined.
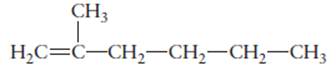
Concept Introduction:
The process of the mixing of orbitals having similar symmetry and energy to results equal number of orbitals is known as hybridization.
The formed orbitals are known as hybrid orbitals.
Carbon belongs to group 4 of the periodic table and its atomic number is 6.
The electronic configuration of carbon is:
In the hybridization, carbon uses 2s and 2p orbitals.
For example:
In case of sp3 hybridization, one 2s and three 2p orbitals of carbon combine to form four sp3 hybrid orbitals.
The electronic configuration of carbon before and after sp3 hybridization is:
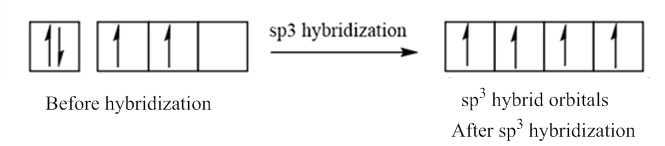
(d)
Interpretation:
The hybridization of each carbon atom in following structure should be determined.
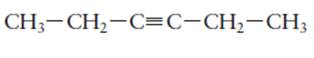
Concept Introduction:
The process of the mixing of orbitals having similar symmetry and energy to results equal number of orbitals is known as hybridization.
The formed orbitals are known as hybrid orbitals.
Carbon belongs to group 4 of the periodic table and its atomic number is 6.
The electronic configuration of carbon is:
In the hybridization, carbon uses 2s and 2p orbitals.
For example:
In case of sp3 hybridization, one 2s and three 2p orbitals of carbon combine to form four sp3 hybrid orbitals.
The electronic configuration of carbon before and after sp3 hybridization is:
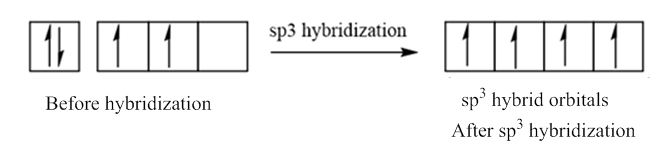
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Principles of Modern Chemistry
- What is the difference between the hybridization of carbon atoms' valence orbitals in saturated and unsaturated hydrocarbons?arrow_forwardHow does the structure of a cycloalkane differ from that of a straight-chain or branched-chain alkane?arrow_forwardWhat is the difference in bonding and in the general molecular formula between an alkene and an alkane with the same number of carbon atoms?arrow_forward
- Write structures for the three isomers of the aromatic hydrocarbon xylene, C6H4(CH3)2.arrow_forwardHow many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





