
Concept explainers
Interpretation: Whether carbocation that is farthest to have an octet in below figure is also least stable and has highest potential energy or not should be determined.
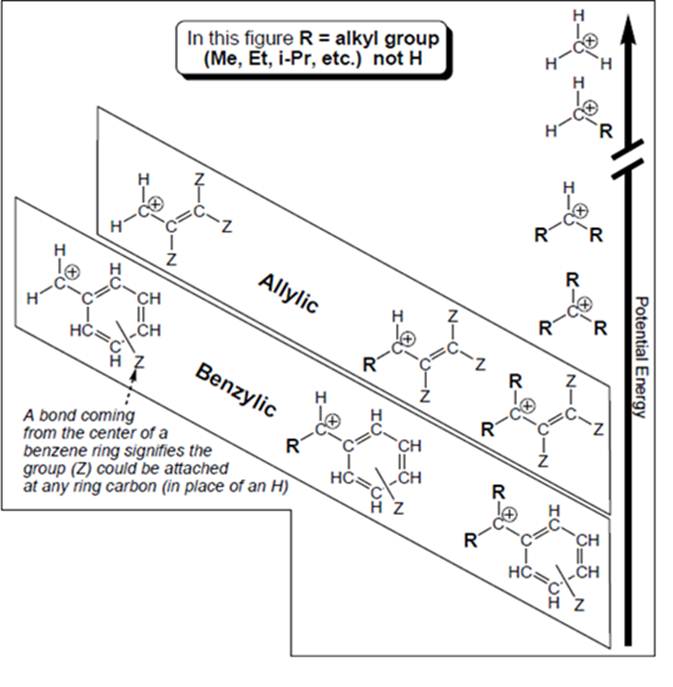
Concept introduction: Organic compounds are covalent in nature that undergoes a reaction by heterolytic cleavage or homolytic cleavage.
In heterolytic cleavage, shared pair of electrons is taken away by one of the atoms which result in charged species. In homolytic cleavage, shared pair of electrons are equally distributed between two atom that results in free radicals.
Carbocation is a general term employed for a postively charged carbon irrespective of valency of carbon. In carbocation, carbon is bonded to 3 atoms or groups and has only sextet of electrons so it behaves as an electron-deficient species. It is
A carbocation is classified as
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
- Explain very detailed why the nitro group add to the ortho of the methyl and not to the Tbutyl group. Please use electron donating and withdrawing groups in the explanation as well as electron density.arrow_forwardGive a clear handwritten answer please with explanation of each moleculearrow_forwardWhich of the dompounds is most basic and why?arrow_forward
- Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. What alkene gives the lowest heat of hydrogenation?arrow_forwardA B ) Which of the above molecules (A or B) have a higher rate of reaction towards aromatic electrophilic reaction? Explain your answer.arrow_forwardGive typing answer with explanation and conclusion What is the major neutral organic product for the following reaction?arrow_forward
- Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. What is/are the product(s) of the ozonolysis of the alkene below?arrow_forwardCompare Electrophilic aromatic substitution reactivity to quinoline and isoquinoline. Please, explain with diagramsarrow_forwardJustify your resposne too...arrow_forward
- Which rxn/s is/are unfavorable? Briefly explain why.arrow_forwardIf there are unshared pairs of electrons on the atoms directly attached to the aromatic ring, they are the groups that provide electrons to the ring with the effect of ……………… and are ………………… directive towards electrophiles. Groups that are double bonded to a more electronegative atom than the atoms directly attached to the ring are ……… steers that withdraw electrons from the ring with the effect of .................. Groups whose electronegativity is higher than the sp2 carbon of the ring directly attached to the ring will withdraw electrons from the ring as ............. and ……… they are routers. Halogens, on the other hand, withdraw electrons from the ring as ………......... due to their high electronegativity and are ……….. directing. A. Resonance/(o-, p-)/ resonance/(m-)/ inductive/(o-, p-)/ inductive/(o-, p-) B. Resonance/(o-, p-)/ resonance/(m-)/ inductive/(o-, p-)/ inductive/(m-) C. Resonance/(o-, p-)/ resonance/(m-)/ inductive/(m-)/ inductive/(o-, p-) D. Inductive/(o-, p-)/…arrow_forwardIt says that this answer is wrong and that the major product structure should contain one or more cyclic (ring) compounds. Reconsider the question description or reaction conditions and try again. Can you help?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
