
Concept explainers
Interpretation: Each carbocation in below figure should be labelled as
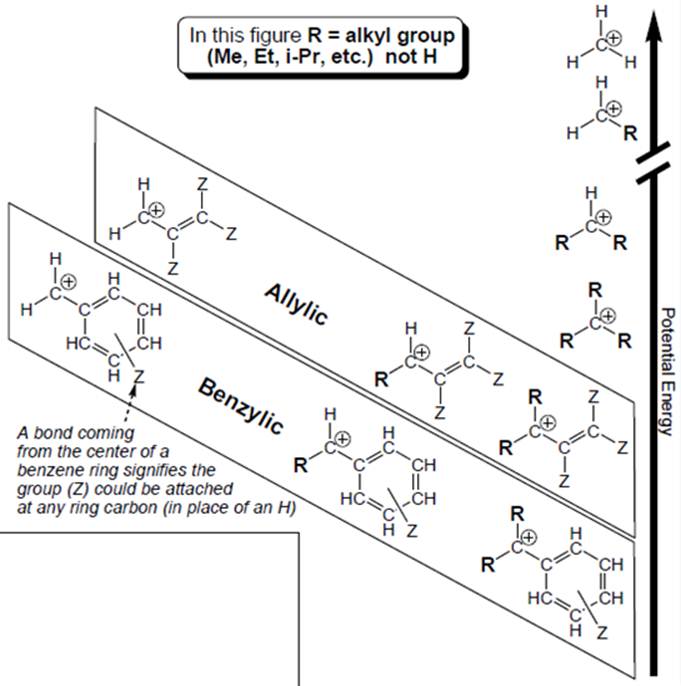
Concept introduction: Organic compounds are covalent in nature that undergoes a reaction by heterolytic cleavage or homolytic cleavage.
In heterolytic cleavage, shared pair of electrons is taken away by one of the atoms which result in charged species. In homolytic cleavage, shared pair of electrons are equally distributed between two atom that results in free radicals.
Carbocation is a general term employed for a postively charged carbon irrespective of valency of carbon. In carbocation, carbon is bonded to 3 atoms or groups and has only sextet of electrons so it behaves as an electron-deficient species. It is
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Custom eBook for Organic Chemistry
- Classify each carbocation as primary, secondary, or tertiary. a. b. 人 C. d.arrow_forwardDraw product F. Select Draw C (C3H5)3P*-CH2arrow_forwardDraw the organic product for each reaction sequence. Remember to include formal charges when appropriate. If more than one major product isomer forms, draw only one. 1. CH,CI, AIC, СН, СІ, AICI, 2. HNO,,H,S0, 1. HNO,,H,SO, 2. CH,CIІ, AICІI,arrow_forward
- By following the direction of curve arrow, draw the final product after the reaction. (a) + BF, (c) HCI (b) + BF, (d) + CH,CH,CH,CH2-Liarrow_forwardAdd curved arrow(s) to draw the final step of the mechanism. A .. 3 + NN OD C 4 H₂C- H BÖH CH3 HO CH3 HỌC-O: :OH CH3arrow_forwardClassify each carbocation as 1°, 2°, or 3°. CH2CH3 a. CH;CH,CHCH,CHa b. с. d. е. CH2arrow_forward
- Decide which carbocation in each pair is more stable.arrow_forwardRank the following carbocations in terms of their energy. A B Highest Energy Lowest Energyarrow_forward3. Circle three allylic carbons in thiamine to the right. How many allylic hydrogens does it have? Why is the indicated carbon not allylic? N thiamine N NH₂ S -OH # allylic Hsarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
