
Concept explainers
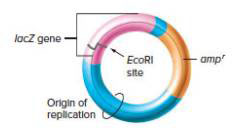
The lacZ gene from E. coli encodes the enzyme β-galactosidase, which can catalyze the conversion of a colorless compound called X-gal into a blue product. Molecular biologists have taken advantage of this property by constructing plasmid vectors that contain the lacZ gene with an EcoRI site in its middle (see figure that follows). After cutting this vector with the EcoRI enzyme, scientists ligate it together with EcoRI-digested human genomic DNA, transform the resultant molecules into ampicillin-sensitive E. coli cells, and plate these cells on petri plates containing ampicillin and X-gal. Some of the colonies growing on this plate are white in color, while others are blue. Why?
Trending nowThis is a popular solution!

Chapter 9 Solutions
Genetics: From Genes To Genomes (6th International Edition)
- The following shows the genotype of a partial diploid bacterial cell - where one chromosomal region containing the lac operon in E,coli is given, and the other fragment is from a plasmid carrying another lac operon from another source. The two are separated by a slash (/). The possible answers indicate with a ʺ+ʺ or a ʺ-ʺ whether β-galactosidase would be expected to be produced at induced levels under two circumstances: 1) first in the absence of lactose and 2) second in the presence of lactose. (Assume that glucose is not present in the medium.)Genotype F: I+ Oc Z-/ Fʹ I- O+ Z+ KEY:I+ = wild-type repressorI- = mutant repressor (unable to bind to the operator)Is = mutant repressor (insensitive to lactose)O+ = wild-type operatorOc = constitutive operator (insensitive to repressor)arrow_forwardThe human hexokinase enzyme has the same function as the bacterial hexokinase enzyme but is somewhat different in its amino acid sequence. You have obtained a mutant bacterial strain in which the gene for hexokinase is missing. If you introduce into your mutant strain a DNA plasmid engineered to contain the DNA coding sequence of the human hexokinase gene, what must you also include? a)The human hexokinase promoter b)The bacterial hexokinase promoter c)Both the human and bacterial promoters d)You cannot engineer a bacteria to produce a human enzymearrow_forwardConsider the following plasmid (size 8000 bp), with restriction sites at the positions indicated: (see image) a) This plasmid is digested with the enzymes listed below. Indicate how many fragments will begenerated in each case, and give the sizes of the fragments.PstIXhoICombination of PstI + XhoI + EcoRI (triple digest) b) Draw the banding pattern you would expect to observe if each of these digestions is loaded into a separate well of an agarose gel, and the fragments separated by electrophoresis. In the first well you load a DNA marker (M) containing fragments with sizes of 1000 bp, 2000 bp, 4000 bp and 8000 bp. c) This gel is transferred to a membrane in a Southern blot experiment, and hybridised to a radioactively labelled 200 bp probe, which anneals to the plasmid DNA at the position indicated on the diagram above. Draw the autoradiographic profile you would expect to observe for the membrane.arrow_forward
- Explain the basis for the following statement: Transcription of two genes on a plasmid can occur without the concomitant action of a topoisomerase, but only if those two genes are oriented in opposite directions.arrow_forwardA plasmid, pUC18, contains the ampicillin-resistance gene, the origin of replication, and the ß - gal gene, which codes for the B-galactosidase protein. This protein can break down the synthetic chemical X-gal, producing a blue product that stains the entire cell blue (but is harmless to the bacteria). At the beginning of the B-gal gene there are several unique restriction sites (some of them are shown in the diagram below). You wish to clone a 1.0-kb Xbal fragment into the pUC18 plasmid, so you cut the plasmid with Xbal and, after removing the enzyme, mix the Xbal-cut plasmid with the 1.0-kb fragment, ligate, and transform competent bacteria. Pati Xbal EcoRI B-gal A Amp ori Figure: pUC18 plasmid map (a) On what medium would you grow your transformed bacteria? (b) Do you expect the bacteria carrying plasmid pUC18 (without the insert) to be blue or white when grown in the presence of X-gal? Explain.arrow_forwardE. coli strains diploid for the lac region were constructed by introducing a plasmid carrying the lac genes. The plasmid carries one copy of the lac region, and the chromosome carries the other copy. The two copies of the lac region have different genotypes, as shown in the chart below. Indicate whether the products of the lacy gene (permease) and the lacZ gene (B-galactosidase) will be inducible, uninducible, or constitutive in each strain (assuming glucose is absent). lac region on plasmid lac region on chromosome permease B-galactosidase I-o+Z+Y- I+o+ Z-Y+ I+o+Z+Y- I+o° Z=Y+ I- oº Z+Y- I+o+ Z-Y+ Is o+ Z+Y- I+o+ Z-Y+ I+ oc Z+Y- IS O+ Z-Y+arrow_forward
- A shuttle vector is a vector (usually a plasmid) constructed so that it can propagate in two different host species. One of the most common types of shuttle vectors is the yeast shuttle vector. Examples of such vectors derived from yeast are Yeast Episomal Plasmid (YEp), Yeast Integrating Plasmid (YIp) and Yeast Replicating Plasmid (YRp). Among these three vectors, YIp has the lowest transformation frequency and copy number per cell. Explain why Ylp is still popularly used despite its limitations.arrow_forwardUsing the plasmid map of pBCH2.0 provided above, predict how many DNA fragments would be formed if this plasmid was digested with restriction enzyme BamHI.arrow_forwardTransposon mutagenesis was used to generate a library of mutants within the Salmonella genome. You are trying to identify a colony with the transposon inserted in the pathogenic related gene SPI-1 using PCR. Forward and reverse primers are generated that flank either side of the gene and yield a wild type product that is 900 bases in length. Which of the colonies sampled in the gel would you expect to contain the SPI-1 gene with transposon insertion? 3,000 2,000 1,000 700 500 300 100 Ladder Colony A Colony B Colony C Colony D Colony E none colonies A&C colonies B&E O colonies A, C, &D colonies B, D, &E -arrow_forward
- In E. coli, the gene bioD+ encodes an enzyme involved in biotin synthesis, and galK+ encodes an enzyme involved in galactose utilization. An E. coli strain that contained wild-type versions of both genes was infected with P1 phage, and then a P1 lysate was obtained. This lysate was used totransduce (infect) a strain that was bioD− and galK−. The cellswere plated on a medium containing galactose as the sole carbonsource for growth to select for transduction of the galK+ gene.This medium also was supplemented with biotin. The resultingcolonies were then restreaked on a medium that lacked biotin tosee if the bioD+ gene had been cotransduced. The following resultswere obtained:What information do you know based onthe question and your understanding of the topic?arrow_forwardIn E. coli, the gene bioD+ encodes an enzyme involved in biotin synthesis, and galK+ encodes an enzyme involved in galactose utilization. An E. coli strain that contained wild-type versions of both genes was infected with P1 phage, and then a P1 lysate was obtained. This lysate was used totransduce (infect) a strain that was bioD− and galK−. The cellswere plated on a medium containing galactose as the sole carbonsource for growth to select for transduction of the galK+ gene.This medium also was supplemented with biotin. The resultingcolonies were then restreaked on a medium that lacked biotin tosee if the bioD+ gene had been cotransduced. The following resultswere obtained:What topic in genetics does this question address?arrow_forwardConsider three genes in E. coli: thr+, ara+, and leu+ (which give the cell the ability to synthesize threonine, arabinose, and leucine, respectively). All three of these genes are close together on the E. coli chromosome. Phages are grown in a thr+ ara+ leu+ strain of bacteria (the donor strain). The phage lysate is collected and used to infect a strain of bacteria that is thr− ara− leu −. The recipient bacteria are then tested on selective medium lacking leucine. Bacteria that grow and form colonies on this medium (leu+ transductants) are then replica-plated on medium lacking threonine and on medium lacking arabinose to see which are thr+ and which are ara+. Another group of the recipient bacteria are tested on medium lackingthreonine. Bacteria that grow and form colonies on this medium (thr+ transductants) are then replica-plated on medium lacking leucine and onto medium lacking arabinose to see which are ara+ and which are leu+. Results from these experiments are as follows:…arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





