
Concept explainers
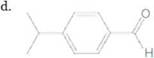
Convert each shorthand structure to a complete structure with all atoms and lone pairs drawn in.
a.
b.
c.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Convert each compound to a skeletal structure. a. CH 3(CH 2) 7CH 3 b. 1,1-diethylcyclohexane c. (CH 3CH 2) 2CHCH 2CH 2CH 3arrow_forwardConvert each shorthand structure to a complete structure with all atoms and lone pairs drawn in. a. (CH3)2CH(CH,),CH3 b. (CH3)3COH c. CH;CO2(CH2)3CH3 ÇI CI d. HO OCH(CH3)2arrow_forwardDraw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a. diethyl ether, (CH3CH2)2O, the first general anesthetic used in medical procedures b.acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon fibers c.dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning products d.acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirinarrow_forward
- . Determine whether the two structures are isomers or the same molecule drawn in two different ways. a. CH3-CH,-c-o-CH3 CH, —С—о—сн, —сн, I b. c. CH3-HC-CH-CH3 ČH;CH3 CH; CH3-HC-CH-CH3 CH3arrow_forwardWhat is the class of organic compound to which the molecule belong? A. H2N B. CH3 HC= CH3 C. CI, CI CI- CH3arrow_forward1. An alkane, P, has the molecular formula, CoHv. An Alkene, Q, has the molecular formula, GHs. a) Name P and Q and write their full structural formula. b) State two differences between P and Q in terms of their structures.arrow_forward
- Name each alkane. a. CH3-CH-CH3 ČH; CH; CH3 b. CH3-CH-CH2-CH-CH2 ČH3 CH, CH, c. CH3-C-C-CH3 ČH, ČH3 CH3 CH, CH2 d. CH3-CH-CH2-CH-CH-CH2-CH2-CH3 ČH3arrow_forwardDraw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a. diethyl ether, (CH;CH2),0, the first general anesthetic used in medical procedures b. acrylonitrile, CH;CHCN, starting material used to manufacture synthetic Orlon fibers C. dihydroxyacetone, (HOCH2)¿CO, an ingredient in sunless tanning products d. acetic anhydride, (CH3CO),0, a reagent used to synthesize aspirinarrow_forwardClassify the following hydrocarbons as alkanes, cycloalkane, alkene, cycloalkenes, alkyne, cycloalkyne, or aromatic.a. CH2 CH CH2b. CH3 C (CH3)2 CH (CH2CH3) CH2 CH2 CH (CH3)2c. (CH3)3 C C C CH (CH3) CH2 CH3arrow_forward
- Identify the circled functional group(s) for each molecule. OH CH3CHCH3 a. ΙΟ ——CH2 b. NH2CH2CH2CH2NHCH3 C. H₂N d. 0 Circle and name all functional group(s) for each molecule CH3 HO OH SH CH2CHCH NHCH(CH3)2 OHarrow_forward1. Identify the circled functional group(s) for each molecule. ОН CH3CHCH3 a. бо =CH₂ b. с. d. NH₂CH₂CH₂CH₂2NHCH3 H₂N 0 CH₂CHCH₂NHCH(CH3)2 стилинен с OHarrow_forward3. Name each of these compounds (including isomer prefixes) and identify the functional groups present. a. H CH CH₂CH/ CH₂ Br b. H CH H El CHSarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
