
Concept explainers
(a)
Interpretation:
The condensed structure of the following structural formula should be determined:
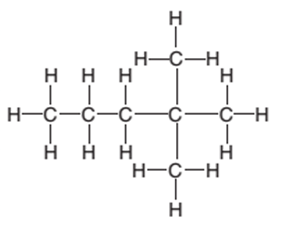
Concept Introduction:
The condensed structural formula of an organic compound represents the atoms of the elements present in the compound and their bonding sequence without any line of a single bond.
(b)
Interpretation:
The condensed structure of the following structural formula should be determined:
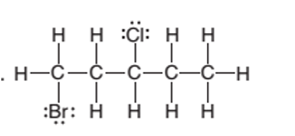
Concept Introduction:
The condensed structural formula of an organic compound represents the atoms of the elements present in the compound and their bonding sequence without any line of a single bond.
(c)
Interpretation:
The condensed structure of the following structural formula should be determined:
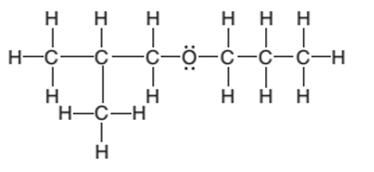
Concept Introduction:
The condensed structural formula of an organic compound represents the atoms of the elements present in the compound and their bonding sequence without any line of a single bond.
(d)
Interpretation:
The condensed structure of the following structural formula should be determined:
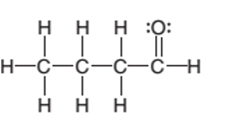
Concept Introduction:
The condensed structural formula of an organic compound represents the atoms of the elements present in the compound and their bonding sequence without any line of a single bond.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Write the condensed structure for each of these skeletal structures. Cl condensed structure: N H condensed structure:arrow_forward2. Convert the following condensed structure into a skeletal structure. CH3CH₂CH(Br)CH₂COCH₂CH(CH₂)2arrow_forward1) Convert the following skeletal structure to a condensed structure. 2) what is the molecular formula of this structure?arrow_forward
- Explain conventions are used for Condensed Structures ?arrow_forwardWrite the condensed structure of 3,4- dimethyl-2-hexenearrow_forwardI have given you a condensed structure. You need to convert it to an accurate bond-line structure. CH3 CH3-CH-CH2-CH-CH₂-C-H 12-CH-CH2₂-C-1 CI Draw (as bond-line structures) isomers of this compound where you only move the chlorine atom. Draw four isomers of the original compound that would have a five carbon chain as the longest chain. [Note: there would be many isomers that will satisfy this. Find any four.] Using the original molecule (and looking at the carbon next to the aldehyde carbonyl) what would be the charge on that carbon if I removed an H atom and left behind the pair of electrons? Circle the best answer Positive Negative Neutral In the space below, draw that structure (from the sentence above) as a bond-line structure. Then, draw a resonance structure for this ion and be sure to add curved arrows to show the movement of electrons.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





