
Concept explainers
(a)
Interpretation:
The following structure of glyceraldehyde should be completed with all H's and lone pairs of atoms:
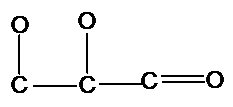
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The atom or group of atoms which are responsible for all special chemical and physical properties of a substance is known as a
(b)
Interpretation:
The following structure of lactic acid should be completed with all H's and lone pairs of atoms:
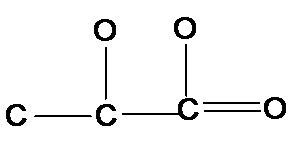
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The atom or group of atoms which are responsible for all special chemical and physical properties of a substance is known as a functional group. These groups are bonded to the parent carbon chain of the organic molecule.
(c)
Interpretation:
The following structure of alanine should be completed with all H's and lone pairs of atoms:

Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The atom or group of atoms which are responsible for all special chemical and physical properties of a substance is known as a functional group. These groups are bonded to the parent carbon chain of the organic molecule.
(d)
Interpretation:
The following structure of dihydroxyacetone should be completed with all H's and lone pairs of atoms:

Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The atom or group of atoms which are responsible for all special chemical and physical properties of a substance is known as a functional group. These groups are bonded to the parent carbon chain of the organic molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- For the following compound identify the lone pairs and indicate if each lone pair is localized or delocalized. Draw out the resonance structures.arrow_forwardConvert each condensed formula to a Lewis structure. CH3(CH2)4CH(CH3)2 (CH3)3CCH(OH)CH2CH3 (CH3)2CHCHO (HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forwardConvert each shorthand structure to a complete structure with all atoms and lone pairs drawn in. a. (CH3)2CH(CH,),CH3 b. (CH3)3COH c. CH;CO2(CH2)3CH3 ÇI CI d. HO OCH(CH3)2arrow_forward
- For highlighted carbon atom , identify which atomic orbitals are used to form each σ bond and which are used to form each π bond.arrow_forwardEach alkane formula listed below has an error in it. Identify and state the error, then rewrite the formula correctly with the same number of carbon atoms. CH5 C2H3 C6H7 CH2Oarrow_forwardDraw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a. diethyl ether, (CH3CH2)2O, the rst general anesthetic used in medical proceduresb. acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon bersc. dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning productsd. acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirinarrow_forward
- Draw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a. diethyl ether, (CH3CH2)2O, the first general anesthetic used in medical procedures b.acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon fibers c.dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning products d.acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirinarrow_forwardButane is an unbranched alkane with the condensed structure CH,CH,CH,CH,. Draw the complete structure of butane. Show all hydrogen atoms. Select Draw Rings More C H Draw the skeletal structure of butane in line-bond (line-angle) mode. Do not show hydrogen atoms. Select Draw Rings More Carrow_forwardDraw an alkane containing one tertiary carbon atom, one secondary carbon atom, and three primary carbon atoms. Show all hydrogen atoms.arrow_forward
- A. Encircle and identify the functional groups of the following molecules. (There may be more than one answer) CH3 2. CHa-с-он ČH3 1. Ibuprofen, a pain reliever : OH CH Ibuprofen 3. Acctone, made by the body when it breaks down fat: H3C `CH3 4. Capsaicin, the pungent substance in chili peppers: Но 5. Arecoline, a veterinary drug used to control worms in animals: ČH3arrow_forward= ORGANIC CHEMISTRY Drawing a skeletal structure from a condensed structure Draw a skeletal ("line") structure of this molecule: OH CH3 -C-CH₂-CH=CH₂ OHarrow_forwardDraw all hydrocarbons that have five C atoms, one triple bond, and no rings (three arrangements).arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





