
Concept explainers
(a)
Interpretation:
To identify the incorrect part present in 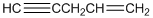 structure.
structure.
Concept Introduction:
The skeletal formula of a compound is also known as shorthand molecular formula. It gives details of its molecules bonding and geometry of molecule.
(b)
Interpretation:
To identify the incorrect part present in 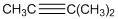 structure.
structure.
Concept Introduction:
The skeletal formula of a compound is also known as shorthand molecular formula. It gives details of its molecules bonding and geometry of molecule.
(c)
Interpretation:
To identify the incorrect part present in 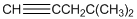 structure.
structure.
Concept Introduction:
The skeletal formula of a compound is also known as shorthand molecular formula. It gives details of its molecules bonding and geometry of molecule.
(d)
Interpretation:
To identify the incorrect part present in the following structure:
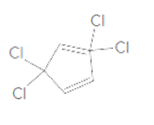
Concept Introduction:
The skeletal formula of a compound is also known as shorthand molecular formula. It gives details of its molecules bonding and geometry of molecule.
(e)
Interpretation:
To identify the incorrect part present in following structure:
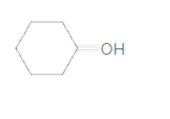
Concept Introduction:
The skeletal formula of a compound is also known as shorthand molecular formula. It gives details of its molecules bonding and geometry of molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- How many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forwardWhat's the missing formula for the following organic compounds?arrow_forwardHi, could someone help me draw these into line structures? Thank you very much.arrow_forward
- Identify all of the functional groupsarrow_forwardFor the following molecule, give, according to IUPAC standards, it's name. Follow the rules for commas, spaces and dashes.arrow_forwardDrawthe characteristic functional group of FOUR of the following six families of organic compounds: Alcohol, Amine, Aldehyde, Ketone, Carboxylic acid, or Ester.arrow_forward
- How many hydrogen atoms are in an alkane moleculewith nine carbon atoms? How many are in an alkenewith nine carbon atoms and one double bond?arrow_forwardWhat are functional groups? Why is it logical and useful to classify organic compounds according to their functional groups?arrow_forwardAre these the same molecule? same molecule isomers different moleculesarrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning




