
Interpretation:
Structure of ciprofloxacin hydrochloride has to be drawn and the
Concept Introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Carboxylic acid: One
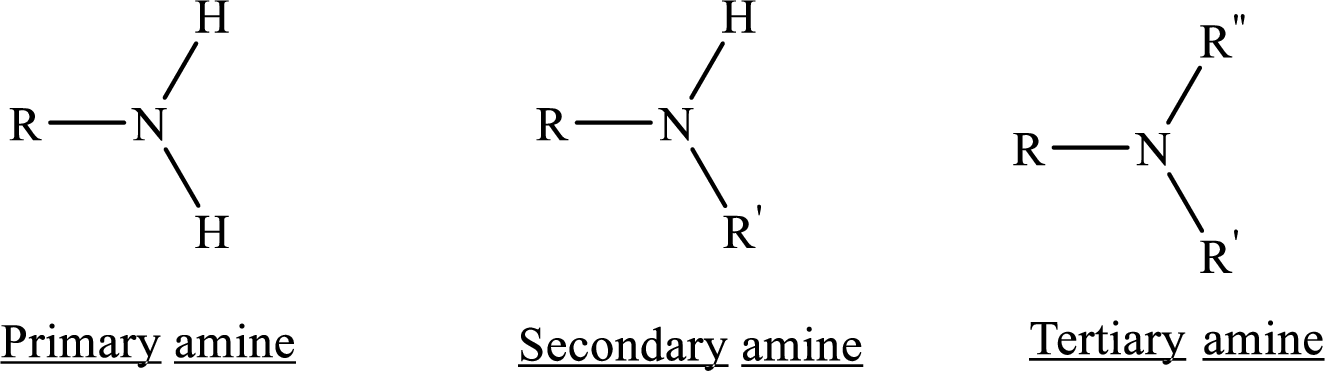
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Chemistry In Context
- What functional groups are present in each molecule? Sucrose C12H22O11 Myristic acid, C14H28O2arrow_forwardYour roommate, a chemistry major, claims to have synthesized the compound CH5 in the lab. Why is that not possible?arrow_forwardWhat 7 functional groups are present in this structure?arrow_forward
- 1. Name the substance, name the group(s) of substances to which this substance belongs. 2. Does this substance contain a functional group or a heteroatom?arrow_forwardIs there any difference between 2-methylpropane and isobutane? Why is the structure in the image not named 2-methylpropane but isobutane. What is meant by "iso"?arrow_forwardIf a research paper appeared reporting the structure of a new molecule with formula C2H8 , most chemists would be highly skeptical. Why?arrow_forward
- drawing of the complete Lewis structure for vitamin b5, showing all atoms, bonds, lone pairs, and charges (as applicable). This structure must be hand-drawn by your group, not a printout from a webpage. c) Identification of all of the organic functional groups for your molecule, clearly labeled on your drawing of its Lewis structurearrow_forwardWhat types of functional groups/bonds does these molecules have?arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





