
(a)
Interpretation:
Structural formula of thioformaldeyde has to be drawn.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
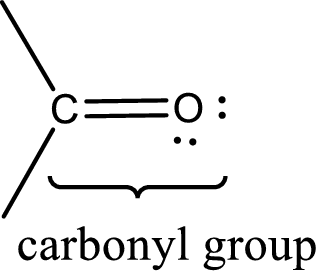
If either of the carbon atom or the oxygen atom present in the carbonyl group is substituted by sulfur atom means it is known as sulfur-containing carbonyl compounds.

(b)
Interpretation:
Structural formula of methanethial has to be drawn.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
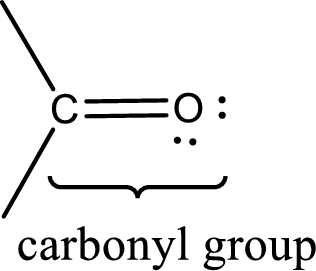
If either of the carbon atom or the oxygen atom present in the carbonyl group is substituted by sulfur atom means it is known as sulfur-containing carbonyl compounds.

(c)
Interpretation:
Structural formula of thioacetone has to be drawn.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
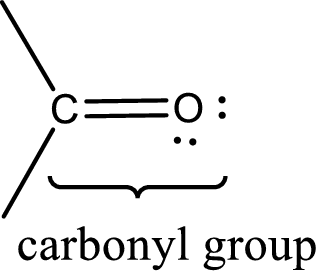
If either of the carbon atom or the oxygen atom present in the carbonyl group is substituted by sulfur atom means it is known as sulfur-containing carbonyl compounds.

(d)
Interpretation:
Structural formula of propanethione has to be drawn.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
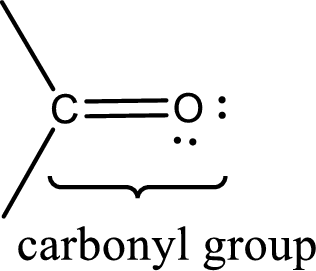
If either of the carbon atom or the oxygen atom present in the carbonyl group is substituted by sulfur atom means it is known as sulfur-containing carbonyl compounds.

Trending nowThis is a popular solution!

Chapter 15 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Drawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forwardIDENTIFY THE FUNCTIONAL GROUP PRESENT IN THESE COMPOUNDS. A = ? B = ? C = ?arrow_forwardThe analgesic phenacetin is synthesized by treating 4- ethoxyaniline with acetic anhydride. a. Which of the following is the structure of 4- ethoxyaniline? OCH₂CH3 NH₂ OCH₂CH3 NH₂ OCH₂CH3arrow_forward
- Draw the enantiomer structure of the following given carbohydrates. Be sure to include appropriate D or L specification.arrow_forwardName the following compound and indicate whether or not is a reducing sugar:arrow_forwardThe explosive trinitrotoluene (TNT) is made by carrying out three successive nitration reactions on toluene. If these nitrations only occur in the ortho and para positions relative to the methyl group, what is the structure of TNT?arrow_forward
- Write the possible structural formulas for all the constitutionally isomeric compounds having the given molecular formula USING THE DASH FORMULA. Take into account the valency of each atom. Indicate the Formal Charge of the Atom, if there is any on the proposed lewis structure. 2. C5H12O (at least 1 ether, at least 1 Alcohol)arrow_forwardwhat is the crystallinity of phenazopyridine? with illustationarrow_forwardConsider these compounds: A. PbBr, B. MnS C. Ag,CO3 D. AIPO, Complete the following statements by entering the letter(s) corresponding to the correct compound(s). (If more than one compound fits the description, include all the relevant compounds by writing your answer as a string of characters without punctuation, e.g, ABC.) Without doing any calculations it is possible to determine that magnesium fluoride is more soluble than and magnesium fluoride is less soluble than| It is not possible to determine whether magnesium fluoride is more or less soluble than by simply comparing Kgp values.arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





