
(a)
Interpretation:
Condensed structural formula for
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
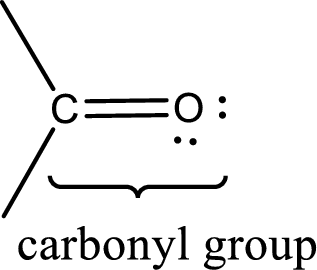
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(b)
Interpretation:
Condensed structural formula for
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
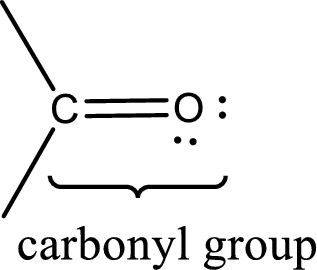
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(c)
Interpretation:
Condensed structural formula for
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
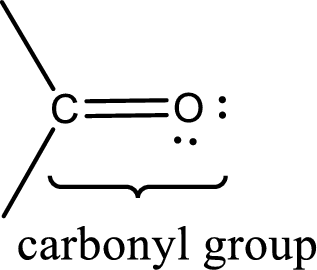
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(c)
Interpretation:
Condensed structural formula for
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
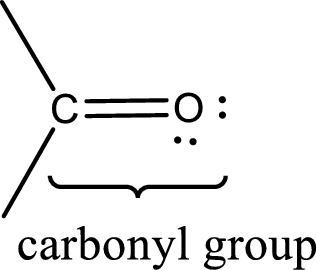
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Listed below are descriptions that may belong to amylose, amylopectin, both, or it may not belong to either. Write A if it describes amylose, B if it describes amylopectin, AB if the statement applies to both, or O if it doesn’t apply to either. 1. Its monosaccharides are bound by at least one α-1,4-glycosidic bond. 2. It contains α-1,6-glycosidic bonds. 3. It can be broken down by the enzyme α-amylase. 4. A polysaccharide made up of gulose. 5. It can form a double helix.arrow_forwardDraw the structure for an unknown disaccharide using the hints given below: Formed with two glucose monomers. The monomers are linked with a B (1 → 6) glycosidic bond. The disaccharide has an anomeric carbon in the a-configuration.arrow_forwardAssign a name to each of the following monosaccharides. Use D and L designations. H b. CH 2OH a. C=0 C=0 HO-C-H H-C-OH HO-C-H Н-С-Он CH,OH CH 2OH a. b. Circle all chiral centers for the monosaccharides shown. С. d. True or False: The molecules shown can rotate plane polarized light?arrow_forward
- a) Use the carbonyl group, number of carbons, and type of stereochemistry (D or L) to classify the following monosaccharides. CH2OH O: CHO I но -H- HO- но -H- HOCH2 HO- -O- CH2OH H- OH CH2OH i.Compound A is a(n with stereochemistry. ii.Compound B is a(n) with stereochemistry.arrow_forwardLook at the structure of stearic acid as well as oleic acid and answer the following questions: a.Write which of these two is saturated fatty acid. b.Which of these two has a higher melting points. c.Which of these two will change the color of bromine water to clear. d.Name the unsaturated fatty acid using both delta and Omega nomenclature I can not separate the question, it is one question with four parts. I only need "D" answered, pleasearrow_forwardNinhydrin is a compound that is commonly used in forensic identification, because it turns purple in contact with the amino acids often found in sweat residue. Using the structure provided, answer the questions below. b. How many carboxylic acid functional groups are present? Explain your answer OH Œ OH 2arrow_forward
- Identify the chiral carbon in each of the following compounds: a. citronellol; one enantiomer has the odor of geranium b. alanine, an amino acidarrow_forwardDraw the structural for the lipid glyceryl tristearate and draw a dotted line about the portion that makes it soluble in nonpolar solvents like cyclohexane. Format BIU ...arrow_forwardDraw the condensed structural formula for the fatty acid whose numerical shorthand designation is 18:2 (△ 9,12)arrow_forward
- Classify the fatty acid with the following structural formula in the ways indicated.a. What is the type designation (SFA, MUFA, or PUFA) for this fatty acid? b. On the basis of carbon chain length and degree of unsaturation, what is the numerical shorthand designation for this fatty acid?c. To which “omega” family of fatty acids does this fatty acid belong? d. What is the “delta” designation for the carbon chain double-bond location for this fatty acid?arrow_forward(a) Label all the O atoms that are part of a glycoside in rebaudioside A. Rebaudioside A, marketed under the trade name Truvia, is a sweet glycoside obtained from the stevia plant, which has been used for centuries in Paraguay to sweeten foods. (b) The alcohol or phenol formed from the hydrolysis of a glycoside is called an aglycon. What aglycon and monosaccharides are formed by the hydrolysis of rebaudioside A?arrow_forwardNinhydrin is a compound that is commonly used in forensic identification, because it turns purple in contact with the amino acids often found in sweat residue. Using the structure provided, answer the questions below. b. How many carboxylic acid functional groups are present? Explain your answer. OH OHarrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





