
Concept explainers
(a)
Interpretation:
Among the three compounds which will react with
Concept Introduction:
In
In organic chemistry, reduction reaction is referred to the number
Alcohols undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give
Aldehyde undergoes oxidation to give carboxylic acid as the product while ketone does not undergo oxidation reaction.
The reverse of
(b)
Interpretation:
The compounds that will react with the Tollen’s solution has to be identified.
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
Alcohols undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Aldehyde undergoes oxidation to give carboxylic acid as the product while ketone does not undergo oxidation reaction.
Tollen’s test:
This is also known as silver mirror test. The reagent that is used in Tollen’s test is silver nitrate and ammonia in water. Aldehyde reacts with Tollen’s reagent, where the silver ion is reduced to silver metal and the aldehyde is oxidized to carboxylic acid.
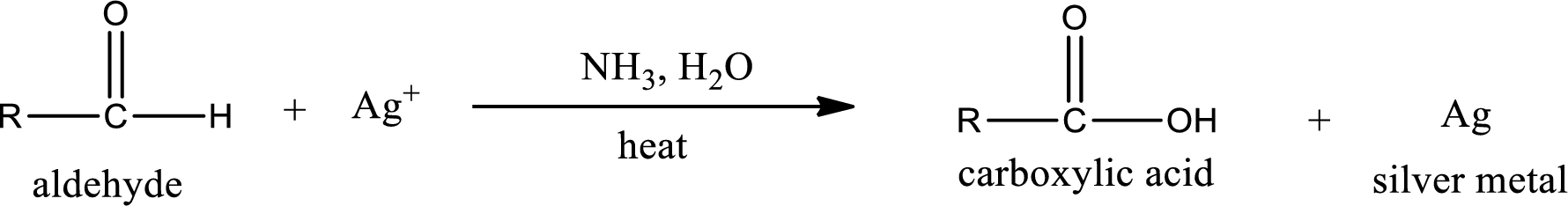
Ketone does not undergo Tollen’s test to deposit silver metal.
(c)
Interpretation:
The compounds that will react with the Benedict’s solution has to be identified.
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
Alcohols undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Aldehyde undergoes oxidation to give carboxylic acid as the product while ketone does not undergo oxidation reaction.
Benedict’s test:
This test is also similar to Tollen’s test. In this test,
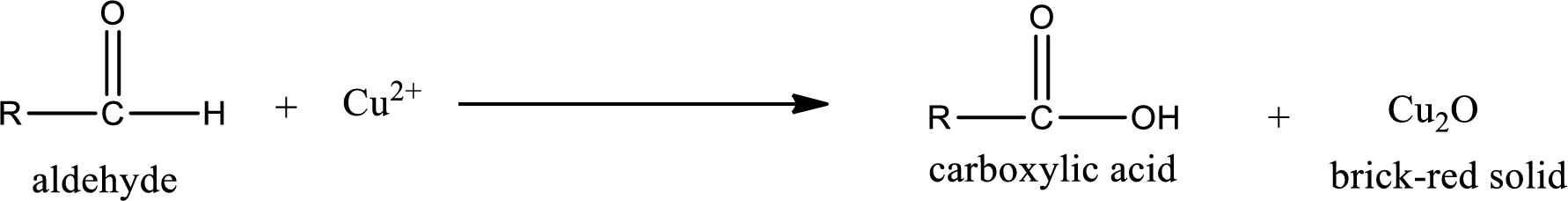
(d)
Interpretation:
Among the three compounds which will react with
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
Alcohols undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Aldehyde undergoes oxidation to give carboxylic acid as the product while ketone does not undergo oxidation reaction.
The reverse of oxidation reaction is reduction reaction. Reduction of aldehyde gives primary alcohol as the product and reduction of ketone gives secondary alcohol as the product. Reduction can be accomplished using hydrogen gas and a metal catalyst namely nickel.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- What is the pH of a cleaning solution with a [H3O*] = 7.4 x 10-10 M H3O*? A. 7.4 B. 9.00 C. 8.13 D. 9.13 E. 7.13arrow_forwardAt 39.9ºC, a solution of ethanol (XetOH = 0.9006, P * etOH = 130.4 Torr) and isooctane (P * iso = 43.9 Torr) forms a vapor phase with YetOH = 0.6667. The total pressure is 185.9 a. Calculate the activity and the activity coefficient of each component.b. Calculate the total pressure the solution would have if it were ideal.c. Comparing the ideal pressure to the actual pressure, what does this indicate about the molecular interactions?arrow_forwardThe analgesic phenacetin is synthesized by treating 4- ethoxyaniline with acetic anhydride. a. Which of the following is the structure of 4- ethoxyaniline? OCH₂CH3 NH₂ OCH₂CH3 NH₂ OCH₂CH3arrow_forward
- A mixture containing glutamic acid, arginine, phenylalanine and valine was subjected to anion exchange chromatography at pH 7.0. Which of the amino acids interacted with the resin (anion exchanger) and last to be eluted with a buffer of decreasing pH? A. Glu B. Val C. Phe D. Argarrow_forwardWhich of the following sulfonamides is used topically in burns? a. Sulfadoxime b. Apsone c. Silver sulfadiazine d. Sulphaethoxazole e. Sulfamethazine f. Sulfametizole g. Phthalylsulfathiazole.arrow_forwardHow many moles of Na2S2O3 would be required to react completely with 0.450 g of AgBr?arrow_forward
- Which of the following substances aren't phenolics? a. lignin b. isoflavones c. isoflavones d. alkaloids e. anthocyaninsarrow_forwardFor this question, use the following reaction and Kc value: 2NH3 (9) * N2(9) + H29) Kc = 3.4 x 104 At equilibrium, what is true about the amount of reactants compared with the amount of products? O A. The amount of reactants are higher than the number of products. B. The number of reactants are lower than the number of products. C. The number of reactants are the same as the number of products. Chparrow_forwardOzone in the upper atmosphere is depleted when it reacts with nitrogen oxides. The rates of the reactions of nitrogen oxides with ozone are important factors in deciding how significant these reactions are in the formation of the ozone hole over Antarctica as shown. One such reaction is the combination of nitric oxide, NO, with ozone, O3:arrow_forward
- You came across an article debating the use of Prevacid® vs. Nexium®. While both drugs have the same therapeutic effect, one is sold as a racemate and the other is sold as a single enantiomer. Previous medications such as Thalidomide and Advair® have sparked a debate about the safety of drugs sold as a racemate. 1. What are Prevacid® vs. Nexium® used to treat?arrow_forwardFill in the blanks: Identify the oxidizing and reducing agent of the given equation: Answers should be the symbol of the element only. 8H*(aq) + 6Cl(aq) + Sn(s) + 4NO3-(aq) SnCl²(aq) + 4NO₂(g) + 4H₂O(1) 1. oxidizing agent 2. reducing agent = Sarrow_forwardWhich molecule is responsible for the blue color in blue essential oils? Choose one answer. a. chamazulene, C15 H16 b. proazulene, C15 H20 c. chamazulene, C14 H16 d. proazulene, C16 H20arrow_forward
