
Hepatocellular carcinoma is the most frequent form of liver cancer. In a patient with heritable hepatocellular carcinoma, formation of the tumor was associated with eight genetic alterations affecting two different oncogenes and three different tumor-suppressor genes. These alterations are:
| i. | Mitotic recombination |
| ii. | A deletion of a chromosomal region |
| iii. | Trisomy |
| iv. | A duplication of a chromosomal region |
| v. | Uniparental disomy (see Fig. 20.24) |
| vi. | A point mutation |
| vii. | Another point mutation |
| viii. | Yet another point mutation |
For parts a–c below, supply all possible correct answers from the preceding list. Remember that the majority of point mutations are loss-of-function mutations.
| a. | Which of the mutations from the preceding list is likely to affect a proto-oncogene? |
| b. | Which of the mutations from the preceding list is likely to involve a tumor-suppressor gene? |
| c. | Which of the mutations from the preceding list involves copy-neutral loss-of-heterozygosity (that is, a loss-of-heterozygosity in which the genomes of the cancerous cells still have two copies of the gene in question, whether or not those copies are functional)? |
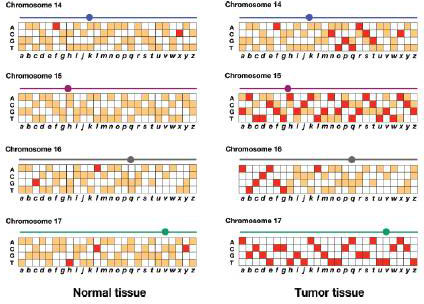
Genomic DNA is prepared from normal white blood cells and from a biopsy of the tumor in this patient. These genomic DNAs are prepared as fluorescent probes that are each hybridized to an ASO microarray of polymorphisms in the human genome (review Figs. 11.16 and 11.17). The results for SNPs a–z on chromosomes 14, 15, 16, and 17 are shown in the accompanying figure. Red and orange represent different levels of fluorescence.
| d. | Based on the microarray data, provide the most accurate localization of the first five types of genetic alterations in the list (i–v). For example, if an alteration involves markers a–e of chromosome 15, write 15a–e. |
| e. | As precisely as possible, indicate the location of the mitotic recombination event involved in the genesis of this cancer |
| f. | If these data allow you to map any of the three cancer-promoting point mutations, provide the most accurate mutation location(s) possible. |
| g. | Of all the genetic alterations i–viii, for which one do you see clear-cut evidence that the mutation or other event was inherited from a parent of the patient? |
| h. | For a tumor-suppressor gene to play a role in cancer, normally both of the copies in the tumor cells must be nonfunctional. For each of the three tumor-suppressor genes contributing to the cancer in this patient, provide a scenario explaining which two hits (i–viii in the list, with vi–viii equivalent) could be responsible, the order in which the hits must have occurred, and whether the hits in question could be inherited or could have occurred somatically. |
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Genetics: From Genes to Genomes
- Describe the steps by which the TP53 gene responds to DNA damage and/or cellular stress to promote cell-cycle arrest and apoptosis. Given that TP53 is a recessive gene and is not located on the X chromosome, why would people who inherit just one mutant copy of a recessive tumor-suppressor gene be at higher risk of developing cancer than those without the recessive gene?arrow_forwardThe dominant condition elliptocytosis causes red blood cells to become misshapen into oval-shaped cells. One of the genes responsible for the abnormal shape encodes the band 4.1 protein that together with ankyrin and other scaffold proteins creates and maintains the spherical concave shape of a normal red blood cell. The gene for band 4.1 protein, EPB41, is found on the p arm of chromosome 1. This is very close to the gene encoding the red blood cell Rhesus (Rh) blood type, either phenotype + (dominant) or - (recessive), with a recombination frequency of 2%. This means that 98% of the time alleles for these two genes are linked and are transmitted together. Diane and Jack are siblings, and both have elliptocytosis and Rh+ blood type. Due to the elliptocytosis, both had emergency splenectomies after having severe anemia. Their younger brother, Devonté, has not yet shown signs of elliptocytosis, but has Rh- blood. André, their dad, also has elliptocytosis and Rh+ blood; while their…arrow_forwardYou are working in a cell biology lab that investigates non-small cell lung cancer cells, which of these cellular features will be suggestive of senescence in the cells observed? Choose all that apply: Group of answer choices Large flattened morphology Reduced incorporation of 5-bromodeoxyuridine (in DNA replication) Increased p53 expression Decreased expression of p15INK4Barrow_forward
- Tumor suppressor genes and oncogenes are implicated in carcinogenesis. However, one can predict whether a gene potentially encodes for a protein that influences carcinogenesis by examining their mutational profile. You sequence the genome of 4 cancers and identify 3 genes of interest. Which of the following genes has the best potential to an oncogene? Tumor 1 Tumor 2 Tumor 3 Tumor 4 Gene A S24F, N465T R33T T345S, G366R P367E, P368Y Gene B S34R, F360I S34R V254I S34E, T67Y Gene C S24F, I322E C255I, E344D S34E, P367Earrow_forwardWhich one of the following events is unlikely to beassociated with cancer?a. mutation of a cellular proto-oncogene in a normaldiploid cellb. a chromosomal translocation with a breakpointnear a cellular proto-oncogenec. deletion of a cellular proto-oncogened. mitotic nondisjunction in a cell carrying a deletionof a tumor-suppressor genee. incorporation of a cellular oncogene into a retroviruschromosomearrow_forwardWhy does a single mutation in a proto-oncogene, turning it into an oncogene potentially lead to a cancerous phenotype, while it takes two mutations in tumor suppressor genes to lead to a cancerous phenotype?arrow_forward
- Chronic myelogenous leukemia is usually associated with which type of chromosome rearrangement? a. Duplication b. Deletion c. Inversion d. Translocationarrow_forwardPlease summarize and make the following paragraph about treacher collins syndrome disease into important bullet points. Thank you!! A mutation in the TCOF1 gene causes up to 93% of Treacher Collins syndrome cases. This gene, which is found on chromosome 5, is in charge of facial developnent. TCOF1 changes spontaneously in around half of all cases at conception, but no one knows what causes the change. In addition, Treacher Collins syndrome is inherited as an autosomal dominant condition in some cases. This indicates that the child inherits the Treacher Collins syndrome gene from one parent and an unaffected copy from the other. Changes in the POLRIC and POLRID genes may be responsible for another 2% of instances. It's unclear how the mutations in the genes create facial abnormalities. However, during weeks three to eight of embryonic development, they are thought to trigger apoptosis (cell death) of facial bone, cartilage, and soft tissue.arrow_forwardThere are three broad categories of cancer-related genes: proto-oncogenes, tumor suppressor genes, and DNA stability/repair genes. Define each of these categories and indicate which one you think the RB1 gene belongs to and why.arrow_forward
- The rb gene encodes a protein that inhibits E2F, a transcriptionfactor that activates several genes involved in cell division.Mutations in rb are associated with certain forms of cancer,such as retinoblastoma. Under each of the following conditions,would you expect the cancer to occur?A. One copy of rb is defective; both copies of E2F are functional.B. Both copies of rb are defective; both copies of E2F arefunctional.C. Both copies of rb are defective; one copy of E2F is defective.D. Both copies of rb and E2F are defective.arrow_forwardThe p53 gene was discovered in 1979, but it was not clear whether the gene functioned as an oncogene or a tumor-suppressor gene. Several years later, researchers showed that both p53 alleles are inactivated in some mouse cancers. This evidence suggests A. the p53 gene is an oncogene because inactivated alleles would produce mutated signal transduction proteins that would result in stimulating cell division. B. the p53 gene is an oncogene because the cell would overproduce transcription factors to compensate for the inactive alleles, resulting in increased cell division. C. the p53 gene is a tumor-suppressor gene because inactivated alleles indicate a loss of protein function which allowed the cancer to develop D. the p53 gene is a tumor-suppressor gene because the cell would produce too few transcription factors for gene activation, resulting in decreased cell division.arrow_forwardResearchers have identified some tumors that have no recurrent mutations or deletions in known oncogenes or tumor-suppressor genes and no detectable epigenetic alterations. However, these tumors often have large chromosomal deletions. What are some possible explanations that could account for the genetic causes behind these tumors?arrow_forward
 Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
