
(a)
Interpretation:
The given material
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in valance band.
Insulator: Insulators are one, in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap.
Conductor: Any material which can conduct electricity is known as conductor. There will be no band gap in metallic conductor and electrons can easily travel.
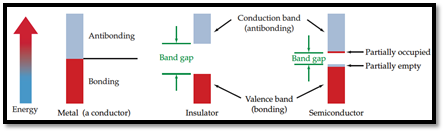
Figure 1
h
(b)
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in valance band.
Insulator: Insulators are one, in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap.
Conductor: Any material which can conduct electricity is known as conductor. There will be no band gap in metallic conductor and electrons can easily travel.

Figure 1
(c)
Interpretation:
The given material white tin has to be classified as a metallic conductor or an n-type semiconductor or a p-type semiconductor and or an insulator.
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in valance band.
Insulator: Insulators are one, in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap.
Conductor: Any material which can conduct electricity is known as conductor. There will be no band gap in metallic conductor and electrons can easily travel.

Figure 1
(d)
Interpretation:
The given material germanium doped with gallium has to be classified as a metallic conductor or an n-type semiconductor or a p-type semiconductor and or an insulator.
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in valance band.
Insulator: Insulators are one, in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap.
Conductor: Any material which can conduct electricity is known as conductor. There will be no band gap in metallic conductor and electrons can easily travel.

Figure 1
(e)
Interpretation:
The given material stainless steel has to be classified as a metallic conductor or an n-type semiconductor or a p-type semiconductor and or an insulator.
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in conduction band.
Insulator: Insulators are one, in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap.
Conductor: Any material which can conduct electricity is known as conductor. There will be no band gap in metallic conductor and electrons can easily travel.

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
General Chemistry: Atoms First
- Sketch the band structure of a Li AND a Si crystal. (ii) How would you expect the electrical conductivity in Li AND Si crystals to vary with temperature? (iii) Account for the fact that the conductivity of Si is enhanced by the addition of small amounts of B.arrow_forwardIs Aluminium is commonly deliberately alloyed with iron? Why ?arrow_forwardClassify as n-type or p-type a semiconductor formed by doping (a) germanium with phosphorus, (b) germanium with indium.arrow_forward
- Account for the relative band gap energies of semiconductors interms of periodic trends?arrow_forwardThe nitride ceramics AIN, GaN, and InN are all semiconductors used in the microelectronics industry. Their band gaps are 580.6, 322.1, and 192.9 kJ/mol, respectively. Which, if any, energies correspond to radiation in the visible region of the spectrum ?arrow_forwardFind melting points of the materials:Aluminum Cast IronLeadMolybdenumNickelPlatinumSilverCarbon SteelStainless SteelTitaniumTungsten ZinBrassAluminum BronzeChromiumCopperGoldInconelarrow_forward
- When you shine light of band gap energy or higher on asemiconductor and promote electrons from the valenceband to the conduction band, do you expect the conductivityof the semiconductor to (a) remain unchanged,(b) increase, or (c) decrease?arrow_forwardDiscuss the need for a high-Tc superconductor.arrow_forwardWhich element produces a p-type semiconductor when doped into silicon?arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning




