
Concept explainers
Interpretation:
The balanced equation for hydrolysis of
Concept introduction:
Sol-gel method:
In this method small molecules are used to prepare solid materials. Sol-gel method converts monomers to sol as colloidal solution which is used as precursor for gel. Mostly used precursors are metal alkoxides. A pictorial representation of sol-gel method is given as follows,
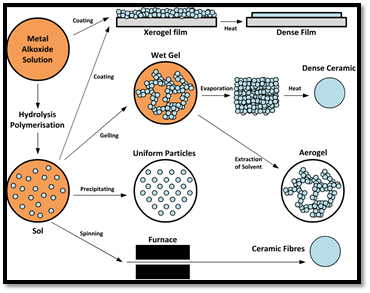
Figure 1
Balanced Chemical equation:
A balanced chemical equation is an equation which contains same elements in same number on both the sides (reactant and product side) of the chemical equation thereby obeying the law of conservation of mass.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
General Chemistry: Atoms First
- Differentiate the properties of carbon filter, ceramics, and micro alloysarrow_forwardIn determining the manganese dioxide content of a pyrolusite mineral. A sample of 0.5261 g of pyrolusite was treated with 0.7149 g of 97.98% sodium oxalate in an acid medium. After the reaction was complete, 30.47 mL of a 0.02160 M potassium permanganate solution was required to titrate for excess unreacted oxalic acid. Calculate the percentage of manganese dioxide in the pyrolusite.arrow_forwardWhat is Oxide Ceramics? Demonstrate the advantages and disadvantages of ceramic materials compared to metals?arrow_forward
- Describe the bonding in calcium oxide, CaO, in terms of bands composed of Ca and O atomic orbitals. How does this model justify the ionic model of this compound?arrow_forwardWhich acid is the best for acid leaching of graphite on the basis of environmental impact ? alkali roasting of graphitearrow_forwardWhat is the role of CO2 in the extractive metallurgy of aluminium from its ore?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


