
Concept explainers
Interpretation:
The ascending order of electrical conductivity of given materials has to be arranged.
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in valance band.
Insulator: Insulators are one in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap. Insulators does not conduct electricity.
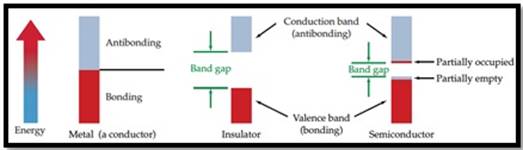
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
General Chemistry: Atoms First
- The following reactions all occur in a blast furnace. Which of these are redox reactions? (a) 3Fe2O3(s)+CO(g)2Fe3O4(s)+CO2(g) (b) Fe3O4(s)+CO(g)3FeO(s)+CO2(g) (c) FeO(s)+CO(g)Fe(l)+CO2(g) (d) C(s)+O2(g)CO2(g) (e) C(s)+CO2(g)2CO(g) (f) CaCO3(s)CaO(s)+CO2(g) (g) CaO(s)+SiO2(s)CaSiO3(l)arrow_forwardSilicon and germanium are both semiconducting elementsfrom Group 4A(14) that can be doped to improve their conduc-tivity. Would each of the following form an n-type or a p-typesemiconductor: (a) Ge doped with P; (b) Si doped with In?arrow_forwardHow many cubic feet of air at a pressure of 760 torr and 0 °C is required per ton of Fe2O3 to convert that Fe2O3 into iron in a blast furnace? For this exercise, assume air is 19% oxygen by volume.arrow_forward
- What causes linkage isomerisation?arrow_forwardWhen aluminum replaces up to half of the silicon atomsin SiO2, a mineral class called feldspars result. The feldspars are the most abundant rock-forming minerals, comprisingabout 50% of the minerals in Earth’s crust. Orthoclase is afeldspar in which Al replaces one-fourth of the Si atoms ofSiO2, and charge balance is completed by K+ ions. Determinethe chemical formula for orthoclase.arrow_forward(a) In the industrial production of iron, what is the reducing substance loaded into the blast furnace? (b) In addition to fur-nishing the reducing power, what other function does this sub-stance serve? (c) What is the formula of the active reducing agent in the process? (d) Write equations for the stepwise reduc-tion of Fe₂O₃ to iron in the furnacearrow_forward
- Would you expect carborundum(SiC) to adopt a diamond structure or one of higher coordination? Explain why?arrow_forwardwhat is the role of silica in the metallurgy of copper?arrow_forwardSketch the band structure of a Li AND a Si crystal. (ii) How would you expect the electrical conductivity in Li AND Si crystals to vary with temperature? (iii) Account for the fact that the conductivity of Si is enhanced by the addition of small amounts of B.arrow_forward
- Several transition metals are prepared by reduction of themetal halide with magnesium. Titanium is prepared by the Krollmethod, in which ore (ilmenite) is converted to the gaseous chlo-ride, which is then reduced to Ti metal by molten Mg. Assuming yields of 84% for step 1 and 93% for step 2,and an excess of the other reactants, what mass of Ti metal canbe prepared from 21.5 metric tons of ilmenite?arrow_forwardGive information about the Nickel Arsenide structure (NiAs). For example: What kind of ionic solid is it, what are its coordination properties, and what is the cation/anion radius ratio? Give information.arrow_forwardEstimate the theoretical density for molybdenum, a BCC metal, when the atomic mass and atomic radius are given by A=95.94 g/mol and R=0.136 nm, respectively. What is the coordination number for Mo atom in a BCC structure?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


