
(a)
Interpretation:
From the given pictures, the material that relates to it has to be identified.
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in conduction band.
Insulator: Insulators are one, in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap.
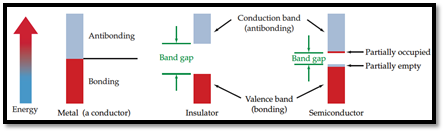
Figure 1
(b)
Interpretation:
The ascending order of electrical conductivity of given materials has to be arranged.
Concept introduction:
Semiconductor: A material which can conduct electricity in range between a metal and an insulator is known as semiconductor. Addition of small amount of impurities increases conductivity of semiconductor and the process is known as doping. n-type and p-type are two types of doped semiconductors.
n-type: This is a type of doped semiconductor in which many free electrons are present in conduction band.
p-type: This is a type of doped semiconductor in which holes are present in conduction band.
Insulator: Insulators in which electrons are filled in valence band with a vacant conduction band and they are parted away by band gap. Insulators does not conduct electricity.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
General Chemistry: Atoms First
- what is the difference between ferromagnetic and paramagnetic?arrow_forwardcalculate the linear electron density of gallium arsenide in with is 10 nm the sub band values of the GeAr are nx =2,ny =2arrow_forwardThe most important oxides of iron are magnetite, Fe3O4,and hematite, Fe2O3. (a) What are the oxidation statesof iron in these compounds? (b) One of these ironoxides is ferrimagnetic, and the other is antiferromagnetic.Which iron oxide is more likely to be ferrimagnetic?Explain.arrow_forward
- Sketch the band structure of a Li AND a Si crystal. (ii) How would you expect the electrical conductivity in Li AND Si crystals to vary with temperature? (iii) Account for the fact that the conductivity of Si is enhanced by the addition of small amounts of B.arrow_forwardIs Aluminium is commonly deliberately alloyed with iron? Why ?arrow_forwardGive information about the Nickel Arsenide structure (NiAs). For example: What kind of ionic solid is it, what are its coordination properties, and what is the cation/anion radius ratio? Give information.arrow_forward
- When aluminum replaces up to half of the silicon atomsin SiO2, a mineral class called feldspars result. The feldspars are the most abundant rock-forming minerals, comprisingabout 50% of the minerals in Earth’s crust. Orthoclase is afeldspar in which Al replaces one-fourth of the Si atoms ofSiO2, and charge balance is completed by K+ ions. Determinethe chemical formula for orthoclase.arrow_forwardExplain how metallic conductors, semiconductors, and insulators are identified and explain their properties in terms of band theory. Why is graphite an electronic conductor and diamond an insulator?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

