
Concept explainers
(a)
Interpretation: The name and structure of the alcohols which on oxidation gives following product
Concept introduction:
Alcohols are the hydrocarbons which have
Alcohols can be of three types on the basis of degree of carbon atom to which the
Primary alcohol: The
Secondary alcohol: The
Tertiary alcohol: The
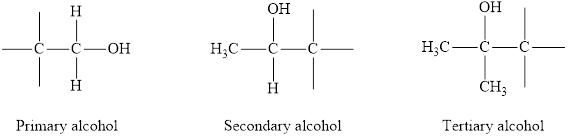
If a strong oxidizing agent is used, the oxidation of primary alcohol gives carboxylic acid.
A weaker oxidizing or partial oxidation of primary alcohol gives aldehydes.
While secondary alcohols always give ketones on oxidation.
(b)
Interpretation: The name and structure of the alcohols which on oxidation gives following product 2-hexanone
Concept introduction:
Alcohols are the hydrocarbons which have
Alcohols can be of three types on the basis of degree of carbon atom to which the
Primary alcohol: The
Secondary alcohol: The
Tertiary alcohol: The

If a strong oxidizing agent is used, the oxidation of primary alcohol gives
A weaker oxidizing or partial oxidation of primary alcohol gives aldehydes.
While secondary alcohols always give
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry & Chemical Reactivity
- Draw and name the five cycloalkane structures of formula C5H10. Can any of these structures give rise to geometric (cis-trans) isomerism? If so, show the cis and trans stereoisomersarrow_forwardGive the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardWrite a condensed structural formula for a dihydroxy compound with the formula C3H8O2.arrow_forward
- Alcohols A, B, and C all have the composition C4H10O. Molecules of alcohol A contain a branched carbon chain and can be oxidized to an aldehyde; molecules of alcohol B contain a linear carbon chain and can be oxidizedto a ketone; and molecules of alcohol C can be oxidized to neither an aldehyde nor a ketone. Write the Lewis structures of these molecules.arrow_forwardwhich functional groups are in C6H12O2 with it having 1 degree of saturation, and is the molecule hexanoic acid?arrow_forwardWrite two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures:(a) ethanol reacts with propionic acid(b) benzoic acid, C6H5CO2H, is added to a solution of sodium hydroxidearrow_forward
- Write a condensed structural formula for a cyclic ketone with the formula C5H8Oarrow_forwardThere are 11 structures (ignoring stereoisomerism) with the formula C4H8O that have no carbon branches. Draw the structures and identify the functional groups in each.arrow_forwardOne mole of an unknown hydrocarbon, compound C, in the presence of a platinum catalyst, adds 98.9 L of hydrogen, measured at 744 mm Hg and 22 degrees C , to form a saturated alkane which contains one ring. When one mole of compound C is reacted with ozone, followed by reduction with (CH3)2S , four moles of only one product was formed, whose condensed molecular formula is CHO -CHO. Give the structure of compound C. Explain your reasoningarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



