
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 24.2, Problem 24.2PP
Interpretation Introduction
Interpretation:
The change in functional group, the utilization of coenzyme and the name of the enzyme for the following reaction needs to be determined.
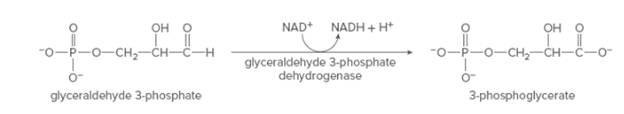 Concept Introduction:
Concept Introduction:
Aerobic respiration occurs in two steps;
- Glycolysis
- Citric acid cycle
Glycolysis is the first step that forms pyruvate. In the presence of oxygen means aerobic respiration, this pyruvate enters in the Krebs cycle and extracts energy in the form of electrons transfer. Electrons are transferred from the pyruvate to the receptors like
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
An enzyme has no activity at an extremely low pH. What could be an explanation for this observation?
a. enzyme inhibition
b. protein denaturation
c. lack of cofactors
d. substrate deactivation
e. no correct response given
The secondary structure of proteins results primarily from which of the following interactions?
a. formation of disulfide linkages
b. formation of hydrogen bonds
c. formation of salt bridges
d. more than one response is correct
e. no correct response
H2 + CH2 = CH2 --> CH3CH3Which of the following could be a substrate for an enzyme that has an induced-fit active site and catalyzes the reaction above?
answer from a to D
a..Which of the enzyme has greater attraction for the subtrate b..Using the graph explain you enzyme in questionC..Which enzyme has greater maximum velocity (VMax)d .why doesnt the reaction rate continue to increase with subtrate concentration?
Chapter 24 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 24.2 - Analyze the following reaction by considering the...Ch. 24.2 - Prob. 24.2PPCh. 24.3 - Prob. 24.1PCh. 24.3 - Prob. 24.2PCh. 24.3 - Prob. 24.3PCh. 24.3 - Prob. 24.4PCh. 24.3 - Prob. 24.5PCh. 24.3 - Prob. 24.6PCh. 24.4 - Prob. 24.7PCh. 24.4 - Prob. 24.8P
Ch. 24.4 - Prob. 24.9PCh. 24.5 - Prob. 24.10PCh. 24.5 - Prob. 24.11PCh. 24.5 - Prob. 24.12PCh. 24.6 - Prob. 24.13PCh. 24.7 - Prob. 24.14PCh. 24.7 - Prob. 24.3PPCh. 24.7 - Prob. 24.15PCh. 24.7 - Prob. 24.16PCh. 24.7 - Use the number of molecules of ATP formed from the...Ch. 24.7 - Prob. 24.18PCh. 24.8 - Prob. 24.19PCh. 24.8 - Prob. 24.20PCh. 24.8 - Prob. 24.21PCh. 24.9 - Prob. 24.4PPCh. 24.9 - What products are formed when each amino acid is...Ch. 24.9 - Prob. 24.22PCh. 24 - Analyze each reaction by considering the...Ch. 24 - Analyze each reaction by considering the...Ch. 24 - Prob. 25PCh. 24 - Prob. 26PCh. 24 - Prob. 27PCh. 24 - Prob. 28PCh. 24 - Prob. 29PCh. 24 - Prob. 30PCh. 24 - Prob. 31PCh. 24 - Prob. 32PCh. 24 - Glucose is completely metabolized to six molecules...Ch. 24 - Why is glycolysis described as an anaerobic...Ch. 24 - Write the overall equation with key coenzymes for...Ch. 24 - Prob. 36PCh. 24 - Prob. 37PCh. 24 - Prob. 38PCh. 24 - Consider the aerobic and anaerobic avenues of...Ch. 24 - Prob. 40PCh. 24 - Prob. 41PCh. 24 - Prob. 42PCh. 24 - Prob. 43PCh. 24 - Prob. 44PCh. 24 - Prob. 45PCh. 24 - Prob. 46PCh. 24 - Prob. 47PCh. 24 - Prob. 48PCh. 24 - Prob. 49PCh. 24 - Prob. 50PCh. 24 - Prob. 51PCh. 24 - Prob. 52PCh. 24 - Prob. 53PCh. 24 - Prob. 54PCh. 24 - Prob. 55PCh. 24 - Prob. 56PCh. 24 - Prob. 57PCh. 24 - Prob. 58PCh. 24 - Prob. 59PCh. 24 - How much ATP is generated by the complete...Ch. 24 - Prob. 61PCh. 24 - Fill in the boxes with the number of moles of each...Ch. 24 - Prob. 63PCh. 24 - Prob. 64PCh. 24 - Prob. 65PCh. 24 - Prob. 66PCh. 24 - Prob. 67PCh. 24 - Prob. 68PCh. 24 - Prob. 69PCh. 24 - Prob. 70PCh. 24 - What is the difference between ketogenic and...Ch. 24 - Prob. 72PCh. 24 - Prob. 73PCh. 24 - Draw the structure of the keto acid formed by the...Ch. 24 - Draw the products formed in each transamination...Ch. 24 - Prob. 76PCh. 24 - Prob. 77PCh. 24 - Prob. 78PCh. 24 - Prob. 79PCh. 24 - Prob. 80PCh. 24 - What metabolic intermediate is formed from the...Ch. 24 - What metabolic intermediate is formed from the...Ch. 24 - Prob. 83PCh. 24 - Prob. 84PCh. 24 - Prob. 85PCh. 24 - Prob. 86PCh. 24 - Prob. 87PCh. 24 - What is the cause of the pain and cramping in a...Ch. 24 - Prob. 89PCh. 24 - Prob. 90PCh. 24 - Prob. 91PCh. 24 - Prob. 92PCh. 24 - Prob. 93PCh. 24 - Prob. 94PCh. 24 - What type of enzyme would catalyze the conversion...Ch. 24 - Prob. 96PCh. 24 - Prob. 97CPCh. 24 - Prob. 98CPCh. 24 - Prob. 99CPCh. 24 - Prob. 100CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- t Plase answer both of youarrow_forwardPhospholipase C was shown to hydrolyze phoshatidylethanolamine and PIP2 with the same efficiency. Both lipids contain the same fatty acyl residues. Will the hydrolysis products of PE and PIP2 have the same effect on protein kinase C? Explain answer!arrow_forwardTwo curves showing the rate versus substrate concentration are shown below for an enzymecatalyzed reaction. One curve is for the reaction in the presence of substance X. The other curve is for data in the absence of substance X. Examine the curves and tell which statement below is FALSE. A. X is an activator of the enzyme. B. The enzyme exhibits non-Michaelis-Menten kinetics. C. X is likely an allosteric effector. D. X is a competitive inhibitorarrow_forward
- Reaction 25.1 Sucrose (aq) + H2O (l) ⇄ Glucose (aq) + Fructose (aq) What is the Keq expression for Reaction 25.1?arrow_forward(please correct answer) A researcher has synthesized an oligopeptide "SLISRAEKLAGVRVGV" and needs to confirm whether the peptides were synthesized with the correct sequence using mass spectrometry. PART A.) What is the sequence of the y6 and b8 ions when the peptide is running tandem MS MS on the structure. PART B.) This peptide is digested with trypsin. What peptide fragments will result?arrow_forwardReaction 25.5 Hb (aq) + Glucose (aq) ⇄ Hb-Glucose (aq) A patient’s blood glucose levels can fluctuate significantly (especially before and after meals). Alternatively, the diabetic A1C marker can be used which measures the percentage of hemoglobin-glucose (Hb-Glucose) in a patient's blood. Using Reaction 25.5, explain why clinicians can use the A1C marker (i.e. levels of Hb-Glucose) in a patient’s blood as a proxy for glucose levels in the blood?arrow_forward
- multiple choice, choose the correct answer When substrate concentration is increased for a simple enzymatic reaction, the reaction order progresses from first-order to zero with respect to substrate concentration because: 1. S-S interactions at high concentrations become strong2. None of the enzyme is found in the ES complex3. Significant product formation results in inhibition of the reaction4. All of the active sites of the enzyme are saturated with substrate at high substrate concentrationarrow_forwardWhich amino acids are converted to oxaloacetate? Select all that apply. A. arginine B. serine C. asparagine D. glutamine E. aspartatearrow_forwardA protein has a chain of 200 amino acids. This is the protein's ________ structure a. primary b. secondary c. tertiary d. quaternary The secondary structure of proteins results primarily from which of the following interactions? a. formation of disulfide linkages b. formation of hydrogen bonds c. formation of salt bridges d. more than one response is correct e. no correct response Protein denaturation results in a disruption of the a. primary structure b. secondary and tertiary structure c. primary and secondary structure d. primary, secondary and tertiary structure e. no correct response is given H2 + CH2 = CH2 --> CH3CH3Which of the following could be a substrate for an enzyme that has an induced-fit active site and catalyzes the reaction above? a. CH3_OH b. CH2=CH_CH3 c. CH3_O_CH2CH3 d. none of the above Enzyme activity is best determined by performing a(n) a. enzyme international unit b. enzyme efficiency test c. enzyme specificity test d. enzyme assayarrow_forward
- 21.78 How will each of the following changes affect the rate of an enzyme-catalyzed reaction whose optimum pH is 7.4? Decreasing the temperature from 37͒C to 5͒C Increasing the temperature from 37͒C to 90͒C Increasing the pH from 7.4 to 10 Increasing the pH from 5 to 7.4 Book: GENERAL ORGANIC AND BIOLOGICAL CHEMISTRY BY SMITH 4TH EDITIONarrow_forwardIn the multienzyme sequence shown below, molecules of D are able to fit to the enzyme E1 and prevent the conversion of A to B. What is this action of E called? AE1BE2CE3D a. effector inhibition b. allosteric inhibition c. feedback inhibition d. competitive inhibition by nonproductarrow_forwardThe following grap shows the relationship between enzyme activity and both pH and temperature for two enzymes (A and B). Based on this graphical information What is the optimum pH for enzyme A? What is the optimum temperature for enzyme B? Which enzyme has the greater activity at a pH of 4? Which enzyme has the greater activity at a temperature of 37.8°C?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY