
(a)
Interpretation:
To draw the product formed when 1-ethylcyclohex-1-ene reacts with HI.
Concept introduction:
The reaction of hydrogen halide like HCl, HBr with an
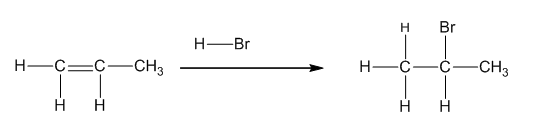
(b)
Interpretation:
To draw the product formed when 1-ethylcyclohex-1-ene reacts with
Concept introduction:
The process hydrogenation is the addition of hydrogen to a double bonded compound in presence of catalyst like Nickel, Platinum etc. The product of this reaction is a saturated compound that is, no single bond present. Therefore,
(c)
Interpretation:
To draw the product formed when 1-ethylcyclohex-1-ene reacts with
Concept introduction:
The reaction of alkene with water in presence of acid gives alcohol. First step in the reaction is protonation and formation of carbocation, Second step is addition of nucelophile (water) to the carbocation and then alcohol forms and regeneration of catalyst takes place.
(d)
Interpretation:
To draw the product formed when 1-ethylcyclohex-1-ene reacts with
Concept introduction:
Reaction of alkene with dihalide gives
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
- The sex attractant of the female tiger moth is an alkane of molecular formula C 18H 38. Is this molecule an acyclic alkane or a cycloalkane?arrow_forwardGive the skeletal structure of the reacting alkene and the reagents the must eb used to produce BrCH2CHOHCH2Clarrow_forwardOn treatment with Cl2/hν, a compound with the formula C9H12 yields only a single monochloride. What is a possible structure of the compound?arrow_forward
- a) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomeric products are formed. Give their structures, and label each asymmetric carbon atom as (R) or (S). What is the relationship between these isomers?arrow_forwardAlkynes undergo many of the same reactions that alkenes do. What organic product(s) would you expect when the following compound is treated with 2 equiv H2, Pd/C?arrow_forward2 methylbutene react with Hbr to givearrow_forward
- Acid-catalyzed hydration of 2-Methyl-1-butene yields two alcohols. The major product does not undergo oxidation, while the minor product will undergo oxidation. Explain why, by showing the structures of theproducts.arrow_forwardAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are the structures of A, B, and C? Write all reactions, and show your reasoning.arrow_forwardCompound A(C7H15Br) is not a primary alkyl bromide. It yields a single alkene(compound B) on being heated with sodium ethoxide in ethanol. Hydrogenation of compound B yields 2,4-dimethylpentane. Identify compounds A and B.arrow_forward
- An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst to give hydrocarbon B. Hydrocarbon A also reacts with OsO4 to give the glycol C. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2COOH, and the other fragment is ketone D (R2C=O). What are the structures of A, B, C and D? Write all reactions.arrow_forwardWhen alkyne A is treated with NaNH2 followed by CH3I, a product havingmolecular formula C6H10O is formed, but it is not compound B. What isthe structure of the product, and why is it formed?arrow_forwardCompounds A and B are isomers of the molecular formula C9H19Br. Both yield the same alkene C in an elimination reaction. Hydrogenation of C yields the product 2,3,3,4 tetramethyl pentane. What are the structures of A, B, and C?arrow_forward
