
Concept explainers
a)
Interpretation: Positive or negative sign above curved arrow to indicate sign of energy change associated with the below process should be determined.
Concept introduction:
b)
Interpretation: Whether bond breakage in
Concept introduction: Chemical reactions that require absorption of energy for their occurrence are known as endothermic reactions. But chemical reactions that are accompanied by the release of energy are known as exothermic reactions. For example, the melting of ice is an example of an endothermic reaction as energy is provided for this process. The reaction between water and calcium chloride is an example of an exothermic reaction.
c)
Interpretation: Whether arrow is more likely to be associated with below left or below right figure should be determined.
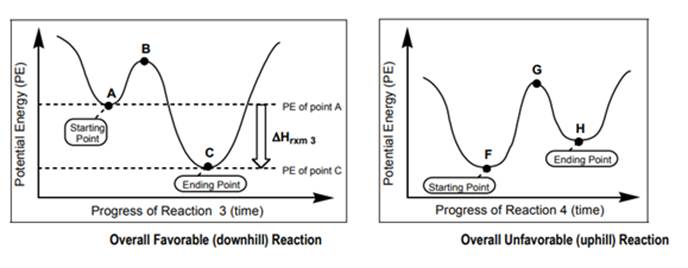
Concept introduction: Chemical reactions that require absorption of energy for their occurrence are known as endothermic reactions. But chemical reactions that are accompanied by the release of energy are known as exothermic reactions. For example, the melting of ice is an example of an endothermic reaction as energy is provided for this process. The reaction between water and calcium chloride is an example of an exothermic reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
