
Concept explainers
(a)
Interpretation:
The most acidic hydrogen should be circled in the following molecule and the amount of energy which is required to remove the most acidic hydrogen should be calculated.
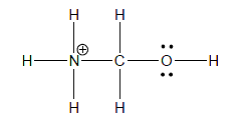
Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A
The value that defines the relationship between the amount of products and reactants at equilibrium is known as equilibrium constant.
(b)
Interpretation:
The expected value of
Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A chemical reaction which takes place between an acid and a base is known as acid-base reaction.
The value that defines the relationship between the amount of products and reactants at equilibrium is known as equilibrium constant.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
- Which is the more stable base? a. Br− or I− b. CH3O− or CH3S− c. CH3CH2O− or CH3COO− d. H2C CH or HC C− e. FCH2CH2COO− or CH2CH2COO- f. CLCH2CH2O- or CL2CHCH2O-arrow_forwardThis reaction makes a carboxylic acid, draw the reaction peoducts for both 1 and 2 (please show reaekson and don't use hend raiting)arrow_forwardFor the following molecules: circle the most acidic hydrogen(s). determine the approximate pKa of the hydrogen you circled rank the compounds from least (1) to most (5) acidic compound.arrow_forward
- Which is a stronger base: RO- or RS-?arrow_forwardSummarize the relationship between pKa and acid strength by completing the following sentences: a. The higher the pKa of an acid, the stronger or weaker the acid. b. The lower the pKa of an acid, the stronger or weaker the acid.arrow_forwardWhich is the stronger base? a. Br- or I- b. CH3O- or CH3S-c. CH3CH2O− or CH3COO−d. H2C =CH e. FCH2CH2COO− or BrCH2CH2COO− f. ClCH2CH2O− or Cl2CHCH2O−arrow_forward
- What is the conjugate base of HS-1? Select one: a. S-2 b. H2S c. H3S+1 d. H2Oarrow_forwardIt’s not A) or B)arrow_forwardNepheliosyne B is a novel acetylenic fatty acid isolated from a New Caledonian marine sponge. (a) Label the most acidic H atom. (b) Which carbon–carbon σ bond is shortest? (c) How many degrees of unsaturation does nepheliosyne B contain? (d) How many bonds are formed from Csp–Csp3? (e) Label each triple bond as internal or terminal.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

