
Concept explainers
Interpretation:The most acidic hydrogen on below structures and reason for the same should be determined.
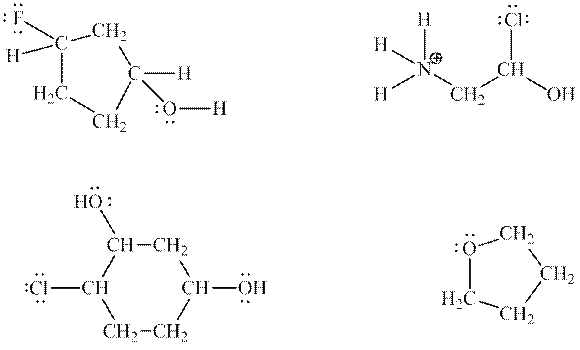
Concept introduction:Electronegative elements have tendency to attract electrons towards it. Since electrons are shared more towards electronegative element, it acquires partial negative charge on it. As a result, other bonded atom develops partial positive charge. Polarization in any molecule arises due to electronegativity difference between bonded atoms. Polarization that is caused by electronegative atom can induce polarization in neighboring bonds to very small extent. This effect is known as inductive effect. Due to this effect, hydrogen attached to positive atom become more acidic than that on neutral atom. This also explains more acidic nature of hydrogens attached to electronegative atoms as compared to other atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
- Consider the following bases: a. For each base above, circle the atom/atoms with the highest PE (will release the most P.E.when a lone pair on this atom combines with an H+ ) b. Rank the bases 1 (highest P.E./strongest base) to 7 (lowest PE/weakest base), and explainyour reasoning.arrow_forwardCircle the most acidic hydrogen atom in the structure shown below.arrow_forwardCircle the most acidic compound in each of the pairs.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
