
Interpretation:
The reason for a solution of 0.10 M HCl to be a better conductor of electricity than 0.10 M CH3COOH must be explained with drawing.
Concept introduction:
HCl is a strong acid and CH3COOH is a weak acid.
Answer to Problem 8E
The sketch of two solutions is shown below depicting ions and molecules.
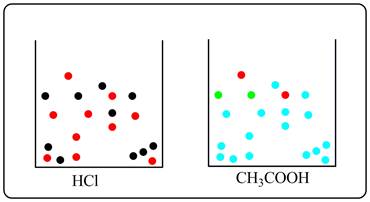
Here, red dots represent H+ ions, black dots represent Cl- ions, green dots represent acetate ions and sky blue dots represent undissociated acetic acid molecules.
Explanation of Solution
From the sketch it is clear that in HCl there is complete dissociation, whereas in acetic acid there are very few molecules of acetic acid which undergoes dissociations. Most of the molecules of acetic acids are undissociated. Thus,number of ions is less in acetic acid. So 0.10 M HCl is a better conductor of electricity.
Chapter U4 Solutions
Living by Chemistry
Additional Science Textbook Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Chemistry: The Central Science (14th Edition)
Chemistry (7th Edition)
Chemistry: The Central Science (13th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
Essential Organic Chemistry (3rd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





