
(a)
Interpretation:TheIR spectra of absorption associated with the C=C bond and vinylic H atoms for the spectral data for 4-methyl-1-pentene needs to be specified in the given figure.
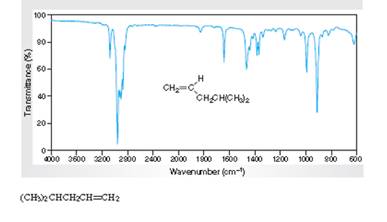
Concept Introduction:
Spectroscopy is the method of identification of structure of the organic molecules with the help of their absorption of certain
(b)
Interpretation: TheH1 spectra for various resonance to the hydrogen nuclei responsible for them needs to be determined.
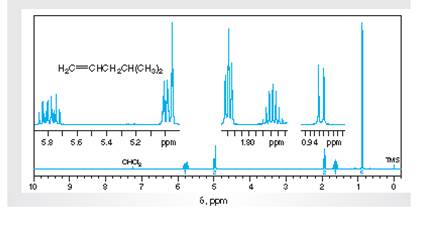
Concept Introduction:
Spectroscopy is the method of identification of structure of the organic molecules with the help of their absorption of certain electromagnetic radiations.
The H1spectroscopy or NMR is based on the nuclear magnetic resonance of radio waves on the H atoms bonded to C atoms present in the organic molecule.
(c)
Interpretation: The C13 spectra for various resonance to the C nuclei responsible for them needs to be determined.
Concept Introduction:
Spectroscopy is the method of identification of structure of the organic molecules with the help of their absorption of certain electromagnetic radiations.
The H1spectroscopy or NMR is based on the nuclear magnetic resonance of radio waves on the H atoms bonded to C atoms present in the organic molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- How do you rationalize the fact that the cyclohexane A value for phenyl (2.8) is bigger than that for isopropyl (2.21)?arrow_forwardA Diels–Alder reaction calls for the use of 5.8 mL5.8 mL of a 4.0 M solution of cyclopentadiene in methanol. Calculate the number of moles of cyclopentadiene present in this volume.arrow_forwardCan you list the possible sources of error that can affect the yield of a Grignard reaction followed by extractions?arrow_forward
- Why is diethyl ether used as a solvent for the reduction reaction of the keystone (3,3-dimethyl-2-butanone) utilizing the reducing agent sodium borohydride (NaBH4). what is the purpose of was repeated washes with water? What is the purpose of repeated washes with brine?arrow_forwardwrite reagents of this reactionarrow_forwardThis experiment is the Separation of a mixture of 3-nitroaniline, Benzoic acid, and naphthalene using an acid-base separation extraction technique What properties of the three compounds in this experiment allow you to use the above technique to separate them?Why are you asked to use multiple portions of the solvent at the extraction steps?Why are you asked not to discard any solvents until you have obtained your products?arrow_forward
- The preparation of Wittig reagents requires a strong base like butyllithium. Sodium methoxide, used in this reaction to prepare the Horner-Emmons-Wittig reagent, is a weaker base than butyllithium. Suggest a reason why this weaker base is sufficient to prepare the Horner-Emmons-Wittig reagent.arrow_forward1. Why is dinitration not a significant process under the conditions used in the Nitration of Bromobenzene experiment. 2. Explain why 4-nitrobromobenzene predominates in the product mixture over 2-nitro benzene when conducting a nitration of Bromobenzene experiment. 3. Using resonance structures, show why no detectable amount of 3-nitrobromobenzene in a nitration of Bromobenzene experiment.arrow_forwardTLC, a powerful analytical tool, can be used to monitor the progress of reactions. The synthesis of ethyl-3-coumarincarboxylate can be monitored by TLC by displaying the starting aldehyde 1, and coumarin product 2, which have very distinct Rf values (Hint: Think about the polarity of compounds 1 and 2 in terms of their abilities to H-bonds to silica gel). What can be determine about the progress of the reaction from analysis of the TLC shown below?arrow_forward
- Reaction with Bromine a. In another test tube, put 10 drops of cyclohexene and 5 drops of bromine and cover the tube with a clean cork b. Shake the tube to mix the contents c. Let it stand for a minute or two to complete the reaction d. Test the gases in the test tube with a piece of blue litmus paper moistened with distilled water and held with tweezers Observations____________________________________ ______________________________________________________________________________________________arrow_forwardWhat procedural error could cause the formation of 1-iodohexane from 1-hexene solutionarrow_forwardwhat is the purpose of addition of acid during the reactionarrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
