
Interpretation:The manner reaction mixture should be analyzed periodically so as to determine the completion of reaction should be outlined.
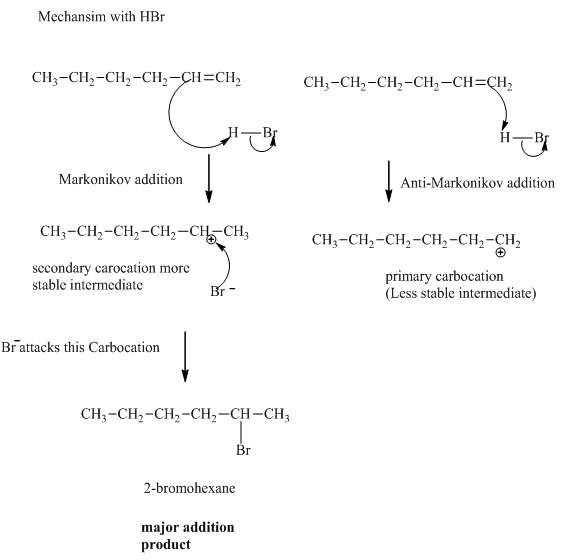
Concept introduction:The general mechanism for the reaction between
The easiest protocol that can be followed to monitor a reaction is TLC analysis.TLC plate is composed of a thin adsorbent layer along with binders such as calcium sulfate for cohesion between adsorbent and the plate. It works on the system of partition equilibrium that involves adsorption to different degrees over the stationary phase of the TLC plate. A suitable solvent system or eluent is chosen that can easily dissolve analytes but not bonds all of them entirely. The solvent system must not be highly polar so as to prevent the affinity of solutes in the mobile phase only.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Give these synthesis mechanism in clear handwritten.arrow_forwardThe phenyl group, C6H5, is known to be an ortho/para-directing group. (a) With that in mind, predict theproduct of the reaction shown here. (b) Justify why it is an ortho/para director by examining the ortho, meta,and para arenium ion intermediates that would beformed during the course of the reaction.arrow_forwardDefine the mechanism of acid–catalyzed halogenation ?arrow_forward
- Fill in Q,R,S,T,U and V with appropriate chemical reagants to make these reactions possible.arrow_forwardwhat are the difference between SN1 and SN2 reaction ? describe the reaction mechanism difference between SN1 and SN2 and give examples with specific detailed explanation.arrow_forwardTLC, a powerful analytical tool, can be used to monitor the progress of reactions. The synthesis of ethyl-3-coumarincarboxylate can be monitored by TLC by displaying the starting aldehyde 1, and coumarin product 2, which have very distinct Rf values (Hint: Think about the polarity of compounds 1 and 2 in terms of their abilities to H-bonds to silica gel). What can be determine about the progress of the reaction from analysis of the TLC shown below?arrow_forward
- Question: How do quantum mechanical effects influence the stability and reactivity of molecules with non-classical carbocations, such as the 2-norbornyl cation, and how does this impact the reaction mechanisms and outcomes?arrow_forwardProvide the mechanism. (Reaction contains oxaphosphentane)arrow_forwardWhat would be a reasonable mechanism for the reaction presented?arrow_forward
- What would be a plausible mechanism for this reactionarrow_forwardQ1: Give one lysis buffer that is commonly used for western blotting experiments and include its components Q3: To make sure that you used a similar amount of samples, what important step should be done before proceeding the electrophoresis stage? Q4. Why is it necessary to store the prepared lysates in a very low temperature?arrow_forwardGive the reaction mechanism and explain in detail. Also perform the woodward Hoffman analysis to explain the selectivity and geometry of the product.arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

