
Concept explainers
(a)
Interpretation: Structure of the major product expected from the ionic addition of
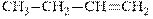
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
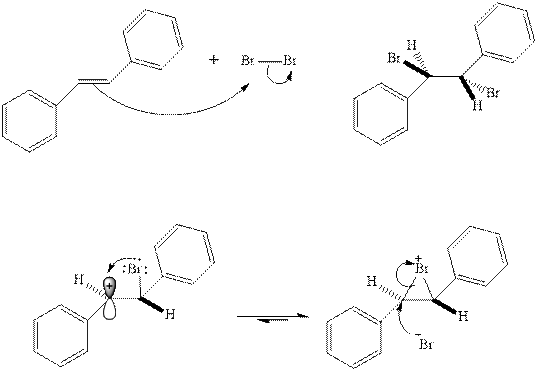
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
(b)
Interpretation: Structure of the major product expected from the ionic addition of
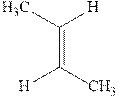
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
(c)
Interpretation: Structure of the major product expected from the ionic addition of
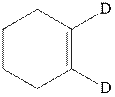
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Define the Mechanism of the Radical Addition of HBr to an Alkene ?arrow_forwardDraw the structural formula for the alkene with molecular formula C5H10 that will react to give the following productsarrow_forwardWrite the steps of the following substitution transformations and draw the structure of the products! (S)-butane-2-ol + cc. HBr →arrow_forward
- Propose the structure of the following: a. An alkane, C6H14 b. A crylic saturated hydrocarbon, C6H12 c. A diene (dialkene), C5H8 d. A keto alkene, C5H8Oarrow_forwardWrite the structure of the compound that will be produced in the following reaction? CH3 –C ≡ C–CH2– CH2 – CH3 + 2HBr→arrow_forwardWrite the structure of the major products in the following reactions and name them.arrow_forward
- Draw the structures of two alkenes that would react to form the haloalkane below upon addition of HBr. Your alkenes should be different regioisomers that yield the haloalkane as the major product without requiring rearrangement to occur.arrow_forwardCompound A(C7H15Br) is not a primary alkyl bromide. It yields a single alkene(compound B) on being heated with sodium ethoxide in ethanol. Hydrogenation of compound B yields 2,4-dimethylpentane. Identify compounds A and B.arrow_forward2-chloropropane is a major product of the reaction of chlorine with propane under ultraviolet light. Write the mechanism for this reaction including the initiation step and the two propagation steps.arrow_forward
- the action of halogenated derivatives of alkanes with KOH: CnH2n + 1 Cl + KOH = CnH2n + 2O + KCl is: a) radical substitution b) nucleophilic substitution c) electrophilic substitution d) addition reactionarrow_forwardName the alkenes that yield 3-methylheptane on catalytic hydrogenation.arrow_forward1. Reaction of 2,3-dimethyl-1-butene with HBr leads to an alkyl bromide, C6H13Br. On treatment of this alkyl halide with KOH in methanol, elimination of HBr occurs and a hydrocarbon that is isometric with the starting alkene is formed. What is the structure of this hydrocarbon and how do you think it is formed from an alkyl bromide? 2. 1-Octen-3-ol is a potent mosquito attractant commonly used in mosquito traps. A number of reactions, including hydrogenation, will transform 1-Octen-3-ol into a less effective molecule. Write a complete reaction equation for the hydrogenation of this alkenol.arrow_forward

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

