
(a)
Interpretation: The structure of the major product formed from alkene indicated should be predicted.
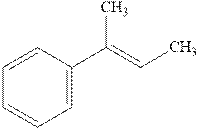
Concept introduction: Hydroboration-oxidation involves the sequence of two reactions. The hydroboration stage involves the treatment of alkene with diborane that generates alkyl borane. In second stage hydrogen peroxide in alkaline medium is used to oxidize the alkyl borane produced in step 1 that leads to the synthesis of alcohols from
Hydroboration is essentially the addition of
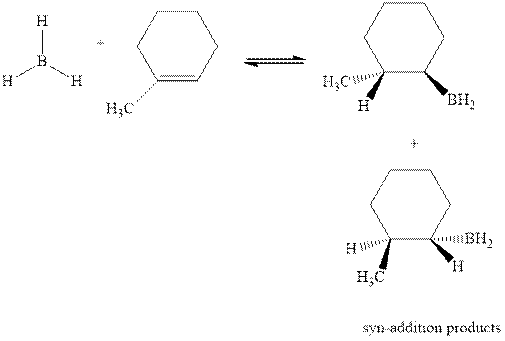
The mechanistic pathway can be illustrated as follows:
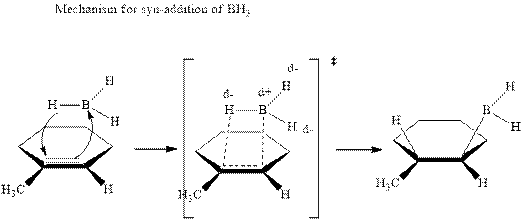
In the first step one equivalent of
(a)
Explanation of Solution
The hydroboration-oxidation productis illustrated as below.
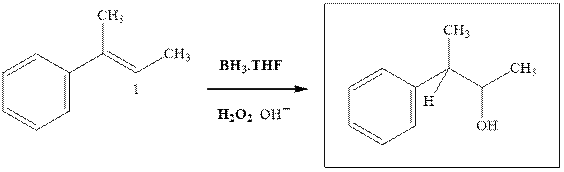
Anti-Markovnikov’s Rule serves as the basis of hydroboration. It states that the negative part of reagent must go to the carbon that has fewer alkyl substituents or more
(b)
Interpretation: The structure of the major product formed from alkene indicated should be predicted.
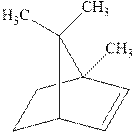
Concept introduction: Hydroboration-oxidation involves a sequence of two reactions. The hydroboration stage involves the treatment of alkene with diborane that generates alkyl borane. In second stage hydrogen peroxide in alkaline medium is used to oxidize the alkyl borane produced in step 1 that leads to the synthesis of alcohols from alkenes.
Hydroboration is essentially the addition of
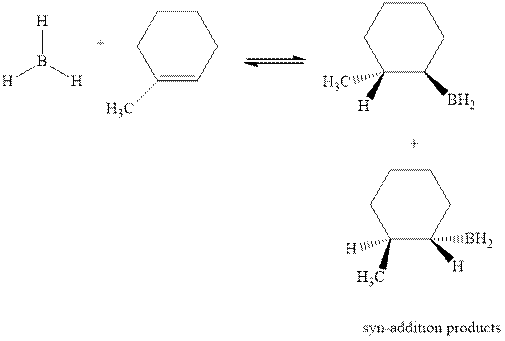
The mechanistic pathway can be illustrated as follows:
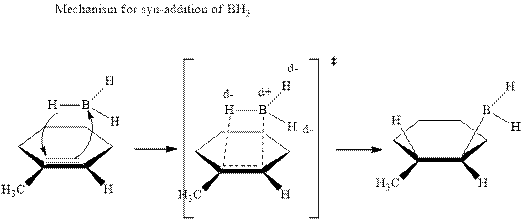
In the first step one equivalent of
(b)
Explanation of Solution
The hydroboration-oxidation product is illustrated as below.

Anti-Markovnikov’s Rule serves as a basis of hydroboration. It states that the negative part of reagent must go to the carbon that has fewer alkyl substituents or more
(c)
Interpretation: The structure of the major product formed from alkene indicated should be predicted.
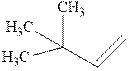
Concept introduction: Hydroboration-oxidation involves a sequence of two reactions. The hydroboration stage involves the treatment of alkene with diborane that generates alkyl borane. In second stage hydrogen peroxide in alkaline medium is used to oxidize the alkyl borane produced in step 1 that leads to the synthesis of alcohols from alkenes.
Hydroboration is essentially the addition of
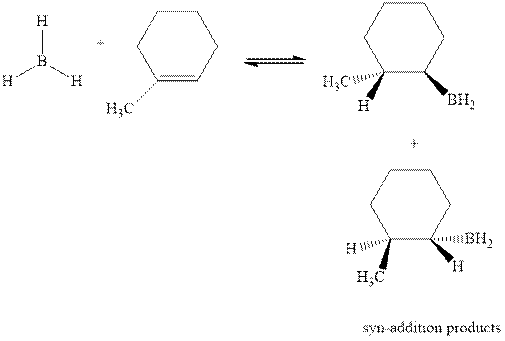
The mechanistic pathway can be illustrated as follows:
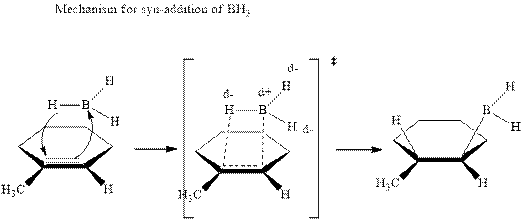
In the first step one equivalent of
(c)
Explanation of Solution
The hydroboration-oxidation product is illustrated as below.
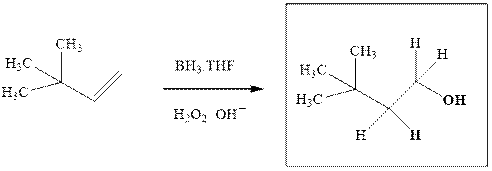
Anti-Markovnikov’s Rule serves as the basis of hydroboration. It states that the negative part of reagent must go to the carbon that has less alkyl substituents or more
Since the terminal olefinic carbon is less substituted
(d)
Interpretation: The structure of the major product formed from alkene indicated should be predicted.
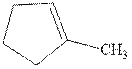
Concept introduction: Hydroboration-oxidation involves a sequence of two reactions. The hydroboration stage involves the treatment of alkene with diborane that generates alkyl borane. In second stage hydrogen peroxide in alkaline medium is used to oxidize the alkyl borane produced in step 1 that leads to the synthesis of alcohols from alkenes.
Hydroboration is essentially the addition of
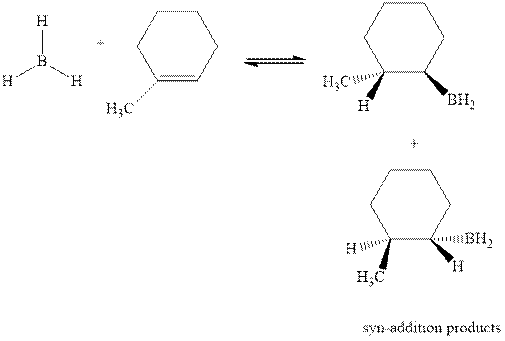
The mechanistic pathway can be illustrated as follows:
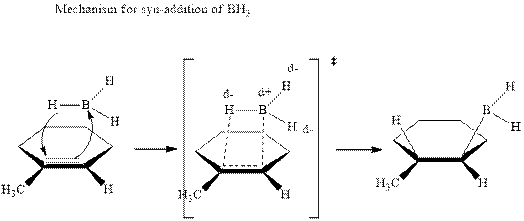
In the first step one equivalent of
(d)
Explanation of Solution
The hydroboration-oxidation product is illustrated as below.

Anti-Markovnikov’s Rule serves as the basis of hydroboration. It states that the negative part of reagent must go to the carbon that has less alkyl substituents or more
(e)
Interpretation: The structure of the major product formed from alkene indicated should be predicted.
Concept introduction: Hydroboration-oxidation involves a sequence of two reactions. The hydroboration stage involves the treatment of alkene with diborane that generates alkyl borane. In second stage hydrogen peroxide in alkaline medium is used to oxidize the alkyl borane produced in step 1 that leads to the synthesis of alcohols from alkenes.
Hydroboration is essentially the addition of
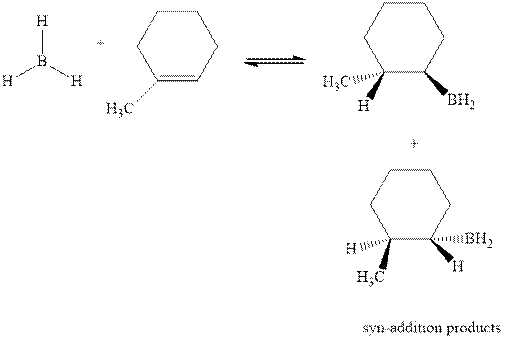
The mechanistic pathway can be illustrated as follows:
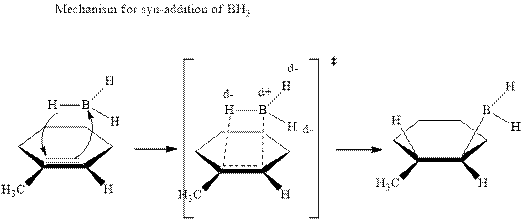
In the first step one equivalent of
(e)
Explanation of Solution
The hydroboration-oxidation product is illustrated as below.

Anti-Markovnikov’s rule serves as the basis of hydroboration. It states that the negative part of reagent must go to the carbon that has less alkyl substituents or more
Want to see more full solutions like this?
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Predict the major products or substrates of the following reactions. If it is possible, write all stereoisomers.arrow_forwardConsider the following statement in reference to SN1, SN2, E1, and E2 reactions of haloalkanes. To which mechanism(s), if any, does the statement apply? Involves a carbocation intermediatearrow_forwardDetermine all of the products obtained from the addition of HCl to the 1,3-diene. Once determined, draw a mechanism that accounts for the formation of every product. Then, Identify and account for the formation of the major adduct/or adducts under these conditions assuming that the reaction is conducted under thermodynamic conditions.arrow_forward
- Addition of HBr to 3,3-dimethyl-1-butene gives a mixture of two isomeric alkyl bromide products. Please daw structures for the two products, and give a mechanistic explanation for their formation.arrow_forwardWrite the mechanisms of the following reactions and examine the products as stereoisomeric.arrow_forwardThere are a variety of isometric 1,2,3,4,5,6-hexachlorocyclohexanes, but one of them does not react with CH3CH2O- to give an elimation product. Provide the structures of this compound and briefly explain why it does not reactarrow_forward
- Provide a reasonable synthetic strategy for the synthesis of a racemic mixture of (1R,2R) and (1S,2S)-2-bromo-1-methylcyclopentanol from the compound shown below... Provide the bond line structure for the major organic products obtained in each step of the proposed strategyarrow_forwardwrite the mechanism and predict the product, and includ the stereochemistryarrow_forwardGive the structures of two different alkyl bromides both of which yield the indicated alkene as the exclusive product of E2 elimination.arrow_forward
- Consider the following statement in reference to SN1, SN2, E1, and E2 reactions of haloalkanes. To which mechanism(s), if any, does the statement apply? Is greatly accelerated in protic solvents of increasing polarityarrow_forwardAccording to the Woodward-Hoffmann rules, is this cycloreversion reaction possible under these conditions?arrow_forwardPredict the Product of the reaction and indicate regiochemistry and stereochemistry when relevantarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





