
Concept explainers
Interpretation:The reason behind capillary tube sealed before the melting point of
Concept introduction:The melting point is usually defined as the particular temperature range that allows liquid phase and solid phase to exist in equilibrium. When a substance melts the temperature remains constant until all the substance has melted down to its liquid form. Melting ranges usually lie at differences of
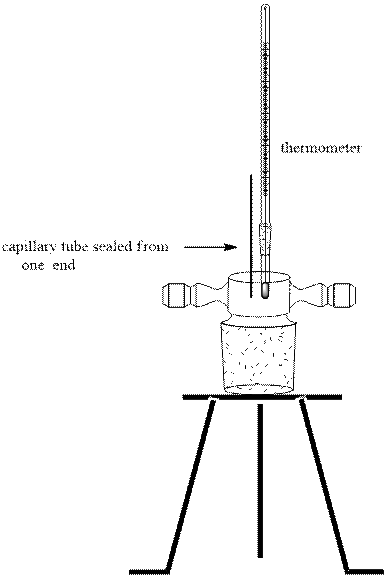
For determination of melting point a fine powder of dried product is made. The capillary tube is sealed from one end and is filled one third by this uniformly smooth powered product. Then this capillary is placed suitable within its position in melting point apparatus and electrical or thermal heat is used to melt the solid. The temperature when the first droplet arises to the complete melt form corresponds to the range of desired product’s melting point. For instance, heat mantle type apparatus for determination of melting pint is illustrated as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Account for the greater amount of internal pressure in the aspirin extraction versus the naphthol extraction ?arrow_forwardWhat is the effect of an insoluble impurity, such as charcoal, on the observed melting point of a compound?arrow_forward1. Why does the solid need to be finely ground and then tightly packed in a melting point capillary tube?arrow_forward
- Re: recrystallization Before packing into a capillary tube, a solid substance has to be powdered. Why is this done on a watch glass rather than on a piece of filter paper?arrow_forwardDraw 4 solvent molecules interacting with either the plate or the analytes and clearly show the relavent intermolecular forces that explain the tlc results.arrow_forwardThe melting point of p-anisalacetophenone is slightly lower than the boiling point of ethanol, during the recrystallization, if you placed the beaker with the crystals on the hot plate without any ethanol, what would have happened to the crystals?arrow_forward
- Diethyl ether and methylene chloride are generally not suitable for use in recrystallization, but are frequently used as extraction solvents. Explainarrow_forwardWhy should the boiling point of the solvent be lower than the melting point of the compound being recrystallized?arrow_forwardWhy can't a mixture of water and diethyl ether be used for recrystallization?arrow_forward
- Some procedures include the addition of sodium carbonate before liquid-liquid extraction. What is the purpose of this step?arrow_forwardWhat are the analyses or instruments that are used to verify or confirm if the extracted substance is indeed caffeine?arrow_forwardCompare the experimental melting point of dibenzalacetone with its literature melting point. What does that tell you?arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

