
a.
Interpretation:
For a given alcohol compound is
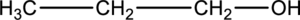
Concept Introduction:
Classification of alcohol:
Generally, the alcohol group is bonded with minimum one alkyl group. The alcohol group is classified into three types, such as primary
b.
Interpretation:
For a given alcohol compound is
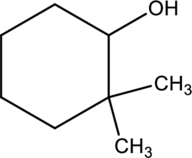
Concept introduction:
Refer to part a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General Organic & Biological Chemistry
- Classify the carbon atoms in each compound as 1°, 2°, 3°, or 4°. a. CH3CH2CH2CH3 b. (CH3)3CHarrow_forwardWhich alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3larrow_forwardWhat products are formed when each alcohol is oxidized with K2Cr2O7? a. CH3CH2CH2CH2CH2OHarrow_forward
- Which of these compounds forms hydrogen bonds with a solvent such as ethanol? 1. CH3CH2CH2OH 2. CH3CH2CH2F 3. CH2OCH2CH3arrow_forwardOrganic Reaction Write an equation for the oxidation of each alcohol. Use [O] above the arrow to indicate an oxidizing agent. If no reaction occurs, write "no reaction" after the arrow. 1. CH3CH2CH2CH2CH2OH CH₂ CH₂CCH₂CH₂ ОН CH CHCH CH CH CH, ОНarrow_forwardDraw the products formed when each alcohol is oxidized with K 2Cr 2O 7. In some cases, no reaction occurs.arrow_forward
- Compound A and compound B (see figure below) are used in different industries. They both have six carbon atoms. Which would be soluble in water and which would be soluble in hexane?arrow_forwardClassify these alcohols as primary (1°), secondary (2°), or tertiary (3°). 1° 2° 3° CH,CH, CH,COH ČH,CH, OH CH,CHCHCH, CH, CH,CH, CH,CCH,OHarrow_forwardH3C. H3C CH3 Br 1-propanolarrow_forward
- 7. Draw a circle around the compound in each horizontal row with the greatest water solubility and draw a rectangle around the compound with the lowest solubility in water. 1-pentanol 1-hexanol 1-octanol methanol OH Но OH Но НО 8. Classify the alcohols below as 1° or 2º or 3º. OH HO HO OHarrow_forward1. Br + CH3O°Na* methanol 2. CI NH3 ethanol 3. CI CH3 O'Na* ethanolarrow_forwardName each aldehyde or ketone. CH, CH, c. CH3-C-CH-CH-CH-C-H ČH,arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





