
Concept explainers
a.
Interpretation:
For the below compound, name has to be determined.
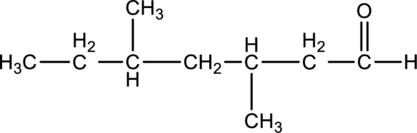
Concept Introduction:
Nomenclature of
Firstly, the
b.
Interpretation:
For the below compound, name has to be determined.
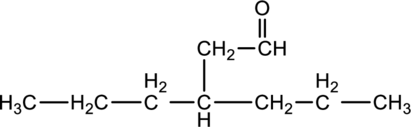
Concept Introduction:
Refer to part “a.”.
c.
Interpretation:
For the below compound, name has to be determined.
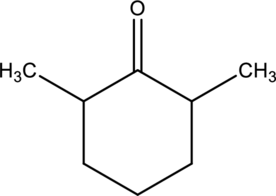
Concept introduction:
Naming of
Firstly, the functional group from the below structure will be identified as ketone and –one is added as the suffix. Subsequently find the longest carbon chain, which is bonded with the ketone group. In case of the cyclic compounds, numbering starts with ketone group attached carbon, which is considered a primary carbon. Prefix changes if any other substituents are present in the structure other than ketone group. In case of more than one substituents naming will be given in alphabetical order.
d.
Interpretation:
For the below compound, name has to be determined.
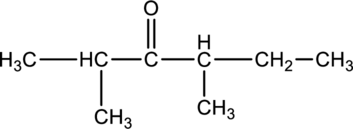
Concept Introduction:
Refer to part “c.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General Organic & Biological Chemistry
- Common names for Aldehydesarrow_forwardName each aldehyde or ketone. CH, CH, c. CH3-C-CH-CH-CH-C-H ČH,arrow_forwardIdentify the most important aldehyde and ketone from Section 14.4 on the basis of amount used, and list at least one characteristic for each that contributes to its usefulness.arrow_forward
- Name each thiol.arrow_forward1. Simple ketones, like acetone, are often used as industrial solvents for many organically based products such as adhesives and paints. They are considered "universal solvents," because they dissolve so many diverse materials. Explain why these chemicals are good solvents.arrow_forward4. Write a general equation representing the preparation of an alcohol by hydrogenation of an aldehyde or a ketone.arrow_forward
- 1. What are the properties of aldehydes and ketones? 2. What will happen to aldehyde and ketones once reduced? Oxidized?arrow_forwardCite some reactions in which formaldehyde behaves differently from other aldehydes.arrow_forwardSelect the correct systematic name of each ester. H3 C- methyl acetate methyl ethanoate ethyl methanoate 2-propanoate H3C-CH2-CH₂- 3-heptanoate propyl butanoate butyl propanoate propyl butanate -CH3 -CH₂-CH2-CH3arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

