
Concept explainers
a.
Interpretation:
For the below ball and stick model, name has to be determined.
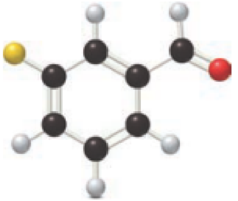
Concept Introduction:
Nomenclature of
Firstly, the functional group from the below structure will be identified as aldehyde and –al is added as the suffix. Subsequently find the longest carbon chain, which is bonded with the -CHO group. In case of the cyclic compounds, numbering starts with –CHO group attached carbon, which is considered as a primary carbon. Prefix changes if any other substituents are present in the structure other than –CHO group. In case of more than one substituents naming will be given in alphabetical order. For the parent
b.
Interpretation:
For the below ball and stick model, name has to be determined
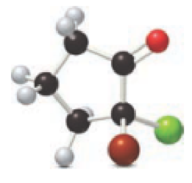
Concept introduction:
Naming of ketones:
Firstly, the functional group from the below structure will be identified as ketone and –one is added as the suffix. Subsequently find the longest carbon chain, which is bonded with the ketone group. In case of the cyclic compounds, numbering starts with ketone group attached carbon, which is considered a primary carbon. Prefix changes if any other substituents are present in the structure other than ketone group. In case of more than one substituents naming will be given in alphabetical order.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General Organic & Biological Chemistry
- 2. What carboxylic acid and alcohol are needed to prepare each ester by Fischer esterification? a) b) c)arrow_forwardDraw the products of each acid–base reaction.arrow_forwardProvide the proper IUPAC name for PhCH₂CH(CH3)CH₂CH₂CHO. 4-methyl-5-phenylpentanol 4-methyl-5-phenylpentanoic acid 4-methyl-5-phenylpentanone 4-methyl-5-phenylpentanalarrow_forward
- Label each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or none of these: a. b. c.arrow_forwardLabel each of the following structures as a hemiacetal, hemiketal, acetal, ketal, or none of these: a. c. b.arrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forward
- Why is it safe for us to consume foods like vinegar that contain acetic acids?arrow_forwardWhat two functional groups react to form the following? a. A hemiacetal b. An acetal c. A ketal d. A hemiketalarrow_forwardIdentify the most important aldehyde and ketone from Section 14.4 on the basis of amount used, and list at least one characteristic for each that contributes to its usefulness.arrow_forward
- 18-18 Propanoic acid and methyl acetate are constitutional isomers, and both are liquids at room temperature. One of these compounds has a boiling point of 141°C; the other has a boiling point of 57°C. Which compound has which boiling point? Explain.arrow_forwardDraw the structure of each compound. p-toluenesulfonic acidarrow_forwardSaponification product of butylpropanoate is: Propanol and Sodium butanoate Butanol and sodium propanoate Butanol and Propanoic acid Butanoic acid and Propanoic acidarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



