
Concept explainers
a.
Interpretation:
For the below ball and stick model, name has to be determined.
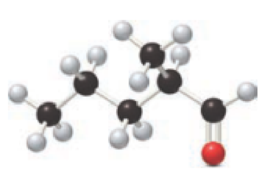
Concept Introduction:
Nomenclature of
Firstly, the functional group from the below structure will be identified as aldehyde and –al is added as the suffix. Subsequently find the longest carbon chain, which is bonded with the -CHO group. In case of the cyclic compounds, numbering starts with –CHO group attached carbon, which is considered a primary carbon. Prefix changes if any other substituents are present in the structure other than –CHO group. In case of more than one substituents naming will be given in alphabetical order. For the parent
b.
Interpretation:
For the below ball and stick model, name has to be determined.
.
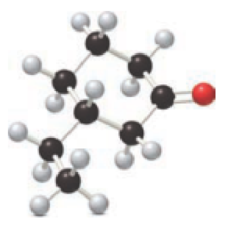
Concept introduction:
Naming of ketones:
Firstly, the functional group from the below structure will be identified as ketone and –one is added as the suffix. Subsequently find the longest carbon chain, which is bonded with the ketone group. In case of the cyclic compounds, numbering starts with ketone group attached carbon, which is considered a primary carbon. Prefix changes if any other substituents are present in the structure other than ketone group. In case of more than one substituents naming will be given in alphabetical order.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General Organic & Biological Chemistry
- Give an acceptable name for each carbonyl compound depicted in the ball-and-stick models.arrow_forwardDraw the products formed when attached alcohol undergoes dehydration with TsOH, and label the major product when a mixture results.arrow_forward2. What carboxylic acid and alcohol are needed to prepare each ester by Fischer esterification? a) b) c)arrow_forward
- Two naturally occurring compounds that contain stable cyclic hemiacetals and acetals are monensin and digoxin. Monensin, a polyether antibiotic produced by Streptomyces cinnamonensis, is used as an additive in cattle feed. Digoxin is a widely prescribed cardiac drug used to increase the force of heart contractions. Label each acetal, hemiacetal, and ether in both compounds.arrow_forwardGive the IUPAC name (including any E,Z designation) for each unsaturated aldehyde.arrow_forwardDraw the structure of a compound fitting each description: a. an aldehyde with molecular formula C,H30 b. a ketone with molecular formula C4HBO c. a carboxylic acid with molecular formula C4H,O2 d. an ester with molecular formula C4H&O2arrow_forward
- Draw the products of each acid–base reaction.arrow_forwardDraw the products formed when attached alcohol undergoes dehydration with TsOH, and label the major product when a mixture results.arrow_forwardLabel each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or none of these: a. b. c.arrow_forward
- Label each of the following structures as a hemiacetal, hemiketal, acetal, ketal, or none of these: a. c. b.arrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forwardWhy is it safe for us to consume foods like vinegar that contain acetic acids?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



