
Concept explainers
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers should be determined.
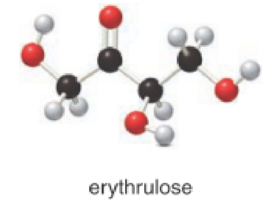
Concept introduction:
Chirality center:
When a carbon atom is bonded to four different groups, then that carbon is named as chiral carbon and it is called as chirality center. Generally, the carbon atom contains tetrahedral valency, therefore it bonded to four atoms or molecules. The chiral center is bonded with four different elements or groups. If the single carbon atom is bonded to two or more identical group or atom, then it is also not considered as chiral carbon. In addition to this, if the carbon atom contains multiple bond then it does not able to bond with four different groups then it is not considered to be as a chirality center.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General Organic & Biological Chemistry
- The structures of 4 isomers of ketopentose are shown. 1) which of the structures are diastereomers of structure A? C, B, or D 2) which of the structures are enantiomers of structure C? B, A, or D 3) which of the structures are stereoisomers of strcuture A?C, D, or Barrow_forwardCellobiose, a disaccharide obtained by the hydrolysis of cellulose, is composed of two glucose units joined together in a 1→4-β-glycoside bond. What is the structure of cellobiose?arrow_forwardPlease draw the expanded structure of D-altrose and its Fischer Projection (chiral C represented as a cross).arrow_forward
- Which compund is optically inactive?arrow_forwardPropose structural formulas for the following polysaccharides. Q.) Pectic acid is the main component of pectin, which is responsible for the formation of jellies from fruits and berries. Pectic acid is a polymer of d-galacturonic acid in the pyranose form joined by a-1,4-glycosidic bonds.arrow_forward4. ) Determine if the following compound is chiral or not. OH HO OH Me ZOH Ме HO HO OH F. CI Ме CI Он Me JCHO CI Br Br CI CH3arrow_forward
- A. Assign each chiral center as R or S B. draw a diastereomer of ephedrine.arrow_forwardA reddish color is obtained when compound A (a disaccharide) is reacted with Benedict solution. Is this compound more likely maltose or sucrosearrow_forwardHow many chiral centers are in B-d-glucopyranose and a-D-galactopyranose? How many stereoisomers of these two aldohexoses can theoretically be drawn?arrow_forward
- Draw the enantiomer and any two diastereomers of cholesterol. Does the OH group of cholesterol occupy an axial or equatorial position?arrow_forwardHow many chirality centres are present in glucose?arrow_forwardAnswer the following questions about the eight aldopentoses: Which form an optically active compound when oxidized with nitric acid?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




