
Chemistry: An Atoms First Approach
2nd Edition
ISBN: 9781305079243
Author: Steven S. Zumdahl, Susan A. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17, Problem 136CWP
Consider a galvanic cell based on the following theoretical half-reactions:
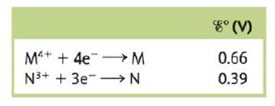
What is the value of ∆G° and K for this cell?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Chemistry: An Atoms First Approach
Ch. 17 - What is a half-reaction? Why must the number of...Ch. 17 - Galvanic cells harness spontaneous...Ch. 17 - Table 17-1 lists common half-reactions along with...Ch. 17 - Prob. 4RQCh. 17 - The Nernst equation allows determination of the...Ch. 17 - Prob. 6RQCh. 17 - Prob. 7RQCh. 17 - Prob. 8RQCh. 17 - What characterizes an electrolytic cell? What is...Ch. 17 - Sketch a galvanic cell, and explain how it works....
Ch. 17 - Prob. 2ALQCh. 17 - Prob. 3ALQCh. 17 - Prob. 4ALQCh. 17 - Sketch a cell that forms iron metal from iron(II)...Ch. 17 - Which of the following is the best reducing agent:...Ch. 17 - Prob. 7ALQCh. 17 - Prob. 8ALQCh. 17 - Explain why cell potentials are not multiplied by...Ch. 17 - What is the difference between and ? When is equal...Ch. 17 - Prob. 11ALQCh. 17 - Look up the reduction potential for Fe3+ to Fe2+....Ch. 17 - Prob. 13ALQCh. 17 - Is the following statement true or false?...Ch. 17 - Prob. 15RORRCh. 17 - Assign oxidation numbers to all the atoms in each...Ch. 17 - Specify which of the following equations represent...Ch. 17 - The Ostwald process for the commercial production...Ch. 17 - Prob. 19QCh. 17 - Prob. 20QCh. 17 - When magnesium metal is added to a beaker of...Ch. 17 - How can one construct a galvanic cell from two...Ch. 17 - The free energy change for a reaction, G, is an...Ch. 17 - What is wrong with the following statement: The...Ch. 17 - When jump-starting a car with a dead battery, the...Ch. 17 - Prob. 26QCh. 17 - Prob. 27QCh. 17 - Consider the following electrochemical cell: a. If...Ch. 17 - Balance the following oxidationreduction reactions...Ch. 17 - Prob. 30ECh. 17 - Prob. 31ECh. 17 - Prob. 32ECh. 17 - Chlorine gas was first prepared in 1774 by C. W....Ch. 17 - Gold metal will not dissolve in either...Ch. 17 - Prob. 35ECh. 17 - Consider the following galvanic cell: a. Label the...Ch. 17 - Prob. 37ECh. 17 - Sketch the galvanic cells based on the following...Ch. 17 - Prob. 39ECh. 17 - Prob. 40ECh. 17 - Prob. 41ECh. 17 - Prob. 42ECh. 17 - Prob. 43ECh. 17 - Give the standard line notation for each cell in...Ch. 17 - Prob. 45ECh. 17 - Prob. 46ECh. 17 - Prob. 47ECh. 17 - Prob. 48ECh. 17 - Prob. 49ECh. 17 - The amount of manganese in steel is determined by...Ch. 17 - Prob. 51ECh. 17 - Prob. 52ECh. 17 - Estimate for the half-reaction 2H2O+2eH2+2OH given...Ch. 17 - Prob. 54ECh. 17 - Glucose is the major fuel for most living cells....Ch. 17 - Direct methanol fuel cells (DMFCs) have shown some...Ch. 17 - Prob. 57ECh. 17 - Using data from Table 17-1, place the following in...Ch. 17 - Answer the following questions using data from...Ch. 17 - Prob. 60ECh. 17 - Consider only the species (at standard conditions)...Ch. 17 - Prob. 62ECh. 17 - Prob. 63ECh. 17 - Prob. 64ECh. 17 - Prob. 65ECh. 17 - Prob. 66ECh. 17 - A galvanic cell is based on the following...Ch. 17 - Prob. 68ECh. 17 - Consider the concentration cell shown below....Ch. 17 - Prob. 70ECh. 17 - The overall reaction in the lead storage battery...Ch. 17 - Prob. 72ECh. 17 - Consider the cell described below:...Ch. 17 - Consider the cell described below:...Ch. 17 - Prob. 75ECh. 17 - Prob. 76ECh. 17 - Prob. 77ECh. 17 - Prob. 78ECh. 17 - Prob. 79ECh. 17 - An electrochemical cell consists of a nickel metal...Ch. 17 - An electrochemical cell consists of a standard...Ch. 17 - Prob. 82ECh. 17 - Consider a concentration cell that has both...Ch. 17 - Prob. 84ECh. 17 - Prob. 85ECh. 17 - Prob. 86ECh. 17 - Consider the following galvanic cell at 25C:...Ch. 17 - Prob. 88ECh. 17 - Prob. 89ECh. 17 - Prob. 90ECh. 17 - Prob. 91ECh. 17 - The solubility product for CuI(s) is 1.1 102...Ch. 17 - How long will it take to plate out each of the...Ch. 17 - The electrolysis of BiO+ produces pure bismuth....Ch. 17 - What mass of each of the following substances can...Ch. 17 - Prob. 96ECh. 17 - An unknown metal M is electrolyzed. It took 74.1 s...Ch. 17 - Electrolysis of an alkaline earth metal chloride...Ch. 17 - What volume of F2 gas, at 25C and 1.00 atm, is...Ch. 17 - What volumes of H2(g) and O2(g) at STP are...Ch. 17 - Prob. 101ECh. 17 - A factory wants to produce 1.00 103 kg barium...Ch. 17 - It took 2.30 min using a current of 2.00 A to...Ch. 17 - A solution containing Pt4+ is electrolyzed with a...Ch. 17 - A solution at 25C contains 1.0 M Cd2+, 1.0 M Ag+,...Ch. 17 - Consider the following half-reactions: A...Ch. 17 - In the electrolysis of an aqueous solution of...Ch. 17 - Copper can be plated onto a spoon by placing the...Ch. 17 - Prob. 109ECh. 17 - Prob. 110ECh. 17 - Prob. 111ECh. 17 - What reaction will take place at the Cathode and...Ch. 17 - Gold is produced electrochemically from an aqueous...Ch. 17 - Prob. 114AECh. 17 - The saturated calomel electrode. abbreviated SCE....Ch. 17 - Consider the following half-reactions: Explain why...Ch. 17 - Consider the standard galvanic cell based on the...Ch. 17 - Prob. 118AECh. 17 - The black silver sulfide discoloration of...Ch. 17 - Prob. 120AECh. 17 - When aluminum foil is placed in hydrochloric acid,...Ch. 17 - Prob. 122AECh. 17 - Prob. 123AECh. 17 - The overall reaction and equilibrium constant...Ch. 17 - What is the maximum work that can be obtained from...Ch. 17 - The overall reaction and standard cell potential...Ch. 17 - Prob. 127AECh. 17 - Prob. 128AECh. 17 - Prob. 129AECh. 17 - Prob. 130AECh. 17 - Prob. 131AECh. 17 - Prob. 132AECh. 17 - Prob. 133AECh. 17 - Prob. 134CWPCh. 17 - Consider a galvanic cell based on the following...Ch. 17 - Consider a galvanic cell based on the following...Ch. 17 - Consider a galvanic cell based on the following...Ch. 17 - An electrochemical cell consists of a silver metal...Ch. 17 - An aqueous solution of PdCl2 is electrolyzed for...Ch. 17 - Prob. 140CPCh. 17 - Prob. 141CPCh. 17 - The overall reaction in the lead storage battery...Ch. 17 - Consider the following galvanic cell: Calculate...Ch. 17 - Prob. 144CPCh. 17 - A galvanic cell is based on the following...Ch. 17 - Prob. 146CPCh. 17 - The measurement of pH using a glass electrode...Ch. 17 - Prob. 148CPCh. 17 - A galvanic cell is based on the following...Ch. 17 - Prob. 150CPCh. 17 - Prob. 151CPCh. 17 - Prob. 152CPCh. 17 - Consider the following galvanic cell: A 15 0-mole...Ch. 17 - When copper reacts with nitric acid, a mixture of...Ch. 17 - The following standard reduction potentials have...Ch. 17 - An electrochemical cell is set up using the...Ch. 17 - Three electrochemical cells were connected in...Ch. 17 - A silver concentration cell is set up at 25C as...Ch. 17 - A galvanic cell is based on the following...Ch. 17 - Prob. 160MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For each reaction listed, determine its standard cell potential at 25 C and whether the reaction is spontaneous at standard conditions. (a) Mn(s)+Ni2+(aq)Mn2+(aq)+Ni(s) (b) 3Cu2+(aq)+2Al(s)2Al3+(aq)+3Cu(s) (c) Na(s)+LiNO3(aq)NaNO3(aq)+Li(s) (d) Ca(NO3)2(aq)+Ba(s)Ba(NO3)2(aq)+Ca(s)arrow_forwardConsider a cell reaction at 25°C where n=4 . Fill in the following table.arrow_forwarda Calculate G for the following cell reaction: Tl(s)Tl+(aq)Pb2+(aq)Pb(s) The Gf for Tl+(aq) is 32.4 kJ/mol. b From G, calculate the standard cell potential for the cell reaction and from this, determine the standard potential for Tl2+(aq)+eTl(s).arrow_forward
- Is it reasonable to conclude that a potential could be assigned to each half-cell in a voltaic cell, based on these data for three voltaic cells? Explain. Zn(s)|Zn2+(aq)||Cu2+(aq)|Cu(s) cell potential=1.10 V Zn(s)|Zn2+(aq)||Ag+(aq)|Ag(s) cell potential=1.56 V Cu(s)|Cu2+(aq)||Ag+(aq)|Ag(s) cell potential=0.46 Varrow_forwardFor each of the reactions, calculate E from the table of standard potentials, and state whether the reaction is spontaneous as written or spontaneous in the reverse direction under standard conditions. (a) Zn(s)+Fe2+(aq)Zn2+(aq)+Fe(s) (b) AgCl(s)+Fe2+(aq)Ag(s)+Fe3+(aq)+Cl(aq) (c) Br2(l)+2Cl(aq)Cl2(g)+2Br(aq)arrow_forwardDetermine the overall reaction and its standard cell potential at 25 C for this reaction. Is the reaction spontaneous at standard conditions? Cu(s)|Cu2+(aq)Au3+(aq)|Au(s)arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Introduction to Electrochemistry; Author: Tyler DeWitt;https://www.youtube.com/watch?v=teTkvUtW4SA;License: Standard YouTube License, CC-BY