
Concept explainers
(a)
Interpretation:
The structure of nitrogen-containing compound that is obtained when the given amide undergoes basic hydrolysis has to be drawn.
Concept Introduction:
Amides are derivatives of
Acidic hydrolysis of amides gives the product as carboxylic acid and
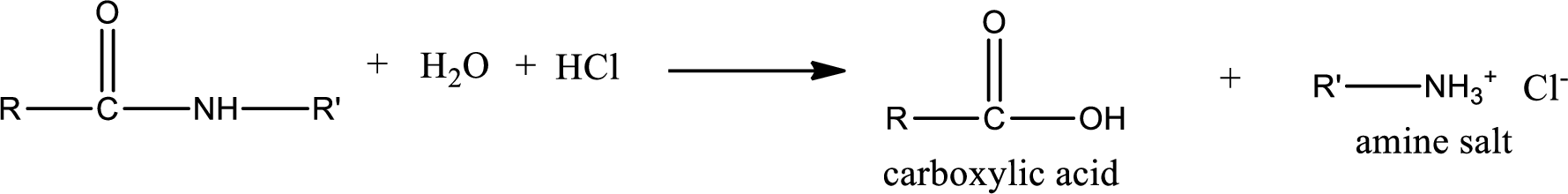
Basic hydrolysis of amides gives the product as carboxylic acid salt and amine. Carboxylic acid salt is obtained because in basic conditions the carboxylic acid is converted into carboxylic acid salt.
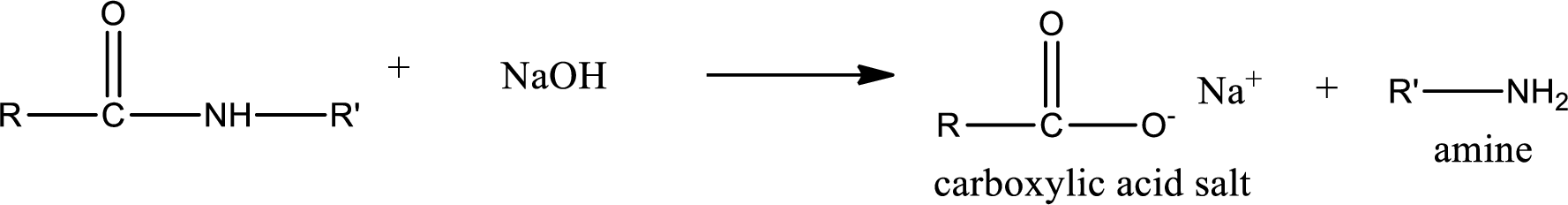
(b)
Interpretation:
The structure of nitrogen-containing compound that is obtained when the given amide undergoes basic hydrolysis has to be drawn.
Concept Introduction:
Amides are derivatives of carboxylic acid. Amides are not much reactive as of carboxylic acids. They are also stable relatively in aqueous solution. But under strenuous conditions amides undergo hydrolysis. The conditions are presence of acid, base or enzymes.
Acidic hydrolysis of amides gives the product as carboxylic acid and amine salt. Amine salt is obtained because in acidic conditions the amine is converted into amine salt.
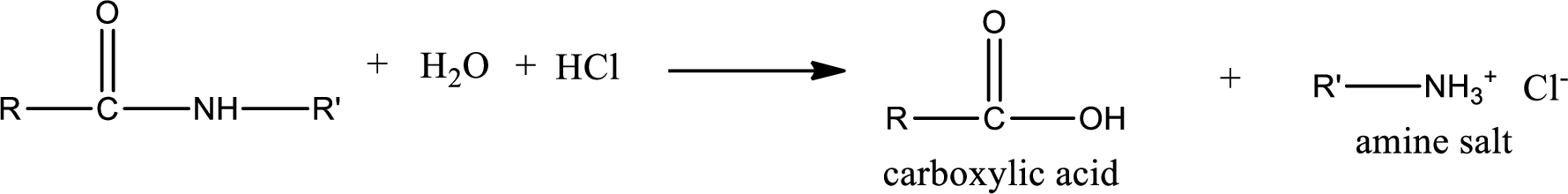
Basic hydrolysis of amides gives the product as carboxylic acid salt and amine. Carboxylic acid salt is obtained because in basic conditions the carboxylic acid is converted into carboxylic acid salt.
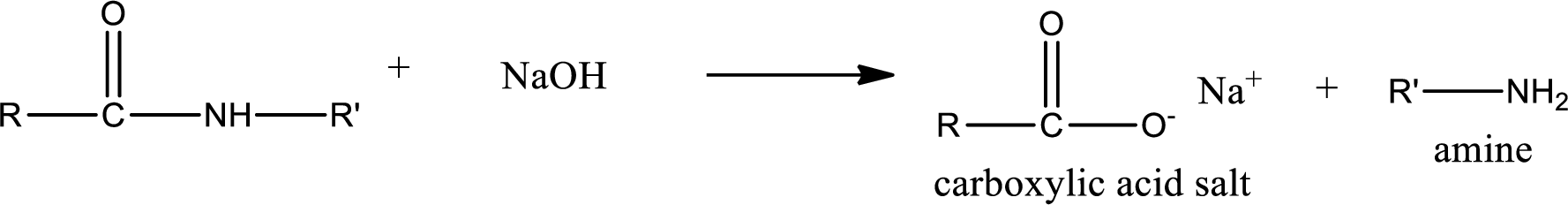
(c)
Interpretation:
The structure of nitrogen-containing compound that is obtained when the given amide undergoes basic hydrolysis has to be drawn.
Concept Introduction:
Amides are derivatives of carboxylic acid. Amides are not much reactive as of carboxylic acids. They are also stable relatively in aqueous solution. But under strenuous conditions amides undergo hydrolysis. The conditions are presence of acid, base or enzymes.
Acidic hydrolysis of amides gives the product as carboxylic acid and amine salt. Amine salt is obtained because in acidic conditions the amine is converted into amine salt.
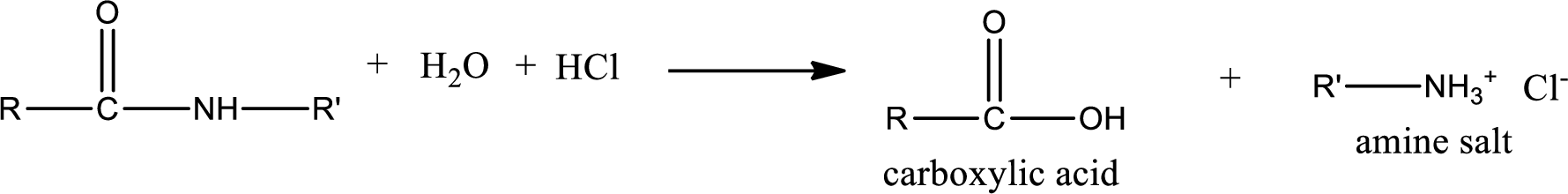
Basic hydrolysis of amides gives the product as carboxylic acid salt and amine. Carboxylic acid salt is obtained because in basic conditions the carboxylic acid is converted into carboxylic acid salt.
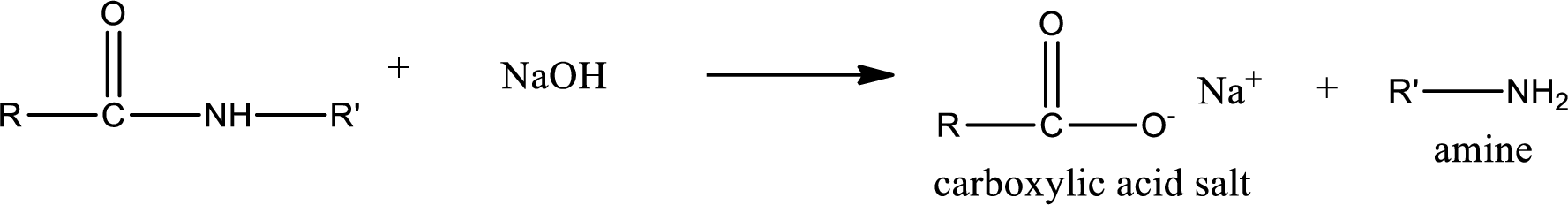
(d)
Interpretation:
The structure of nitrogen-containing compound that is obtained when the given amide undergoes basic hydrolysis has to be drawn.
Concept Introduction:
Amides are derivatives of carboxylic acid. Amides are not much reactive as of carboxylic acids. They are also stable relatively in aqueous solution. But under strenuous conditions amides undergo hydrolysis. The conditions are presence of acid, base or enzymes.
Acidic hydrolysis of amides gives the product as carboxylic acid and amine salt. Amine salt is obtained because in acidic conditions the amine is converted into amine salt.
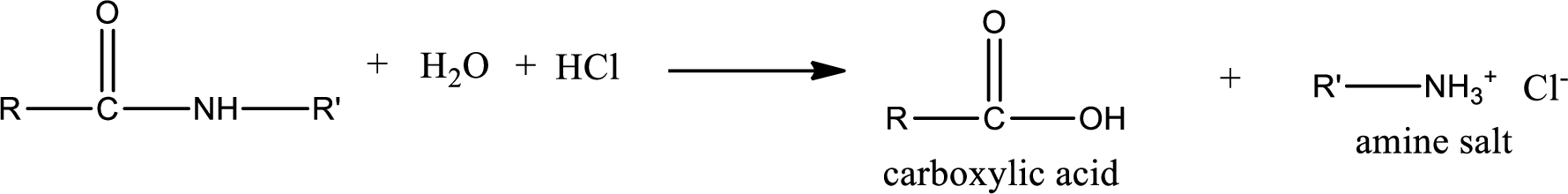
Basic hydrolysis of amides gives the product as carboxylic acid salt and amine. Carboxylic acid salt is obtained because in basic conditions the carboxylic acid is converted into carboxylic acid salt.
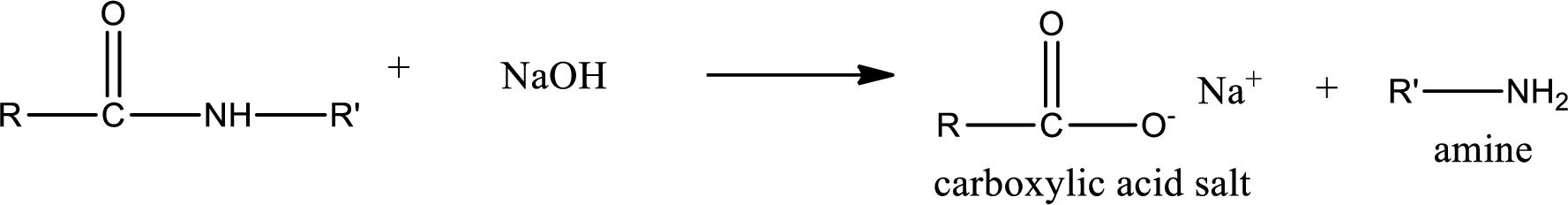
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Match the following structural composition of each polysaccharide with its identity Linear homoglycan of glucose connected by α1-4 linkages Linear sulfated chains of alternating β-D-galactopyranose and 3,6-anhydro- α-galactopyranosyl units Poly-β-Dmannopyranosyluronic acid and/or Poly-α-L-gulopyranosyluronic acid Repeating Poly-D-galacturonic acid residues deacetylated straight-chain amino-polysaccharide polymer linked in a β(1-4) type of linkage a. Alginate b. amylose c. chitosan d. carrageenan e. Pectinarrow_forwardCarbohydrates Instructions: (A) Show the conversion from Fischer to Haworth projections. (B) Draw and give the systematic names of the two (2) possible Haworth structures for the following monosaccharides. 1.D- Ribose 2. D- Galactose 3. D- Fructosearrow_forwardIf thymine nucleotides are degraded by the same enzymes as those catabolizing uridine nucleotides, give the structure of the thymine metabolite that corresponds to B-ureidopropionate.arrow_forward
- Glutathione is a commonly occurring tripeptide; it has considerable physiological importance because it is a scavenger for oxidizing agents. gamma-glutamyl-L-cysteinylglycine. Draw the structure of Glutathione and explain why the systematic name of Glutathione is such.arrow_forwardGive one specific sample structure (Fischer Projection or Haworth) of the following carbohydrate derivatives: - Alditols - Deoxy-sugararrow_forwardCarbohydrates Instructions: (A) Show the conversion from Fischer to Haworth projections. (B) Draw and give the systematic names of the two (2) possible Haworth structures for the following monosaccharides. 1. D- Galactosearrow_forward
- In the monosaccharide derivatives known as sugar alcohols, the carbonyl oxygen is reduced to a hydroxyl group. For example, D-glyceraldehyde can be reduced to glycerol. However, this sugar alcohol is no longer designated D or L. Why?arrow_forwardDraw the two possible Haworth structures (both alpha and beta anomers) for the following monosaccharides and give their corresponding systematic names. [Show the stepwise process]arrow_forwardCarbohydrates Instructions: (A) Show the conversion from Fischer to Haworth projections. (B) Draw and give the systematic names of the two (2) possible Haworth structures for the following monosaccharides. 1. D- Fructosearrow_forward
- Aspartame (NutraSweet), an artificial sweetener, is a dipeptide composed of Asp-Phe in which the carboxyl terminus is modified by the attachment of a methyl group. Draw the structure of Aspartame at pH 7.arrow_forwardExplain why the indole nitrogen of tryptophan is more weakly basic than one of the imidazole nitrogens of histidine What is the relationship between (R)-cysteine and (S)-alanine Do they have the opposite three-dimensional configuration (as the names might suggest) or the same configuration? Is (R)-cysteine a D-amino acid or an L-amino acid?arrow_forwardDraw the structural formula for ⍺-D-glucosyl-(1 6)-D-mannosamine, does the compound have a reducing sugar? If yes, circle the part of this structure that makes the compound a reducing sugar.arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





