
Concept explainers
Interpretation:
On acidic hydrolysis of amide, the organic products that are obtained has to be chosen from the given options.
Concept Introduction:
Amides are derivatives of
Acidic hydrolysis of amides gives the product as carboxylic acid and
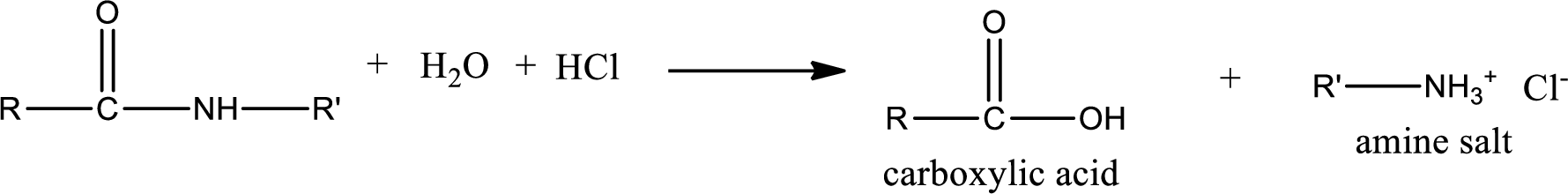
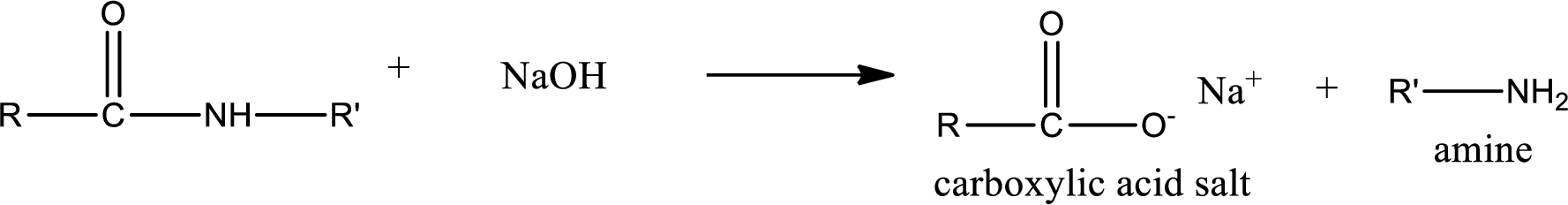
Basic hydrolysis of amides gives the product as carboxylic acid salt and amine. Carboxylic acid salt is obtained because in basic conditions the carboxylic acid is converted into carboxylic acid salt.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Which of the following compounds would be most soluble in carbon tetrachloride, CCl4? a. NH3 d. CH3OHb. H2O e. C6H6c. HClarrow_forwardPeptide bonds are formed via what type of reaction? A. Condensation B. Hydrolysis C. Oxidation-Reduction D. None of the abovearrow_forwardCarnitine combines with fatty acids groups to form acyl carnitine through carnitines ____ group thereby forming a ____ bond a. hydroxyl, acid anhydride b. Carboxylic acid, acid anhydride c. Hydroxyl, ester D. Carboxylic acid, esterarrow_forward
- What is the conjugate base of the weak acid, CH3COOH if it reacts with H2O in an acid-base reaction? A. H3O* B. CH3COO С. ОН- D. H2O E. CH3COOarrow_forwardWhat type of linkage is catalyzed by peptidyl transferase activity? a. anhydride b. mixed anhydride c. amide d. esterarrow_forwardWhat is the isoelectric point of casein? Please choose one correct answer only. A. pH 6.6 B. pH 5.6 C. pH 4.6 D. pH 3.6 E. None of the given optionsarrow_forward
- If we attached an amine group (NH₂) to Carbon 4, what type of amine will be the result? A. Primary amine B. Secondary amine C. Tertiary amine D. Quaternary aminearrow_forward1. which IR stretches and bends indicate what type of functional group we have (alcohol, carboxylic acid, ester), 2. how you can then use IR to monitor the change from the starting materials to the products. 3. indicate and discussion the absorptions that are relevant to alcohol OH and C-O, carboxylic acid OH, carboxylic acid C=O, ester C=O, and C-O). the picture below is an ir spectra of ethanolarrow_forwardCoenzyme A binds to an acetyl group via a __________ linkage. A. carboxylic acid ester B. carboxylic acid anhydride C. phosphoric anhydride D. thioesterarrow_forward
- 59. Arrange the following five hydrocarbons in order of increasing of boiling pointA. 2-methylhexane B. heptane C. 3,3-dimethylpentandeD. Hexane E. 2-methylpentancearrow_forwardIf you heat Holiday “mulled (spiced) wine” to 40oC for 1 hour, you would expect that: a. it would become champagne b. it would contain fewer Calories per gram than celery c. more alcohol would have evaporated than water during the heating process, lowering the alcohol content (the %ABV), because ethanol has a lower boiling point than water d. A1 casein would be converted to A2 casein e. it will give you “a warm Holiday glow”arrow_forwardAnswer the following questions concerning sulfathiazole below by filling ieach blank with the appropriate response. N S: sulfathiazole The hybridization of the nitrogen atom in sulfathiazole is A. sp B. sp² C. sp³ B. sp2 A. sp C. sp3arrow_forward
