
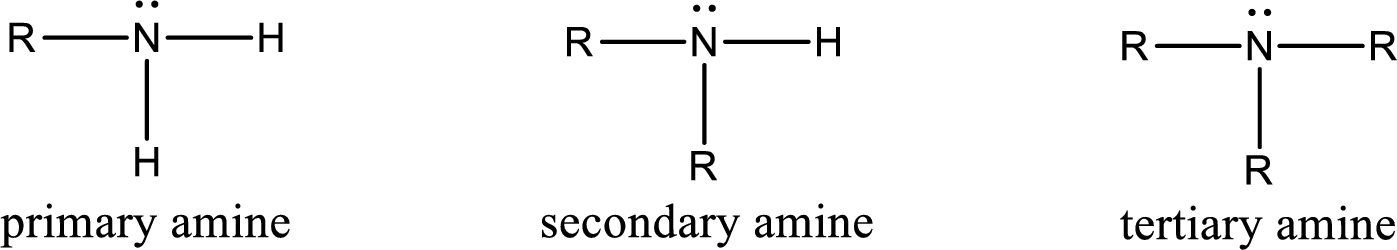
(a)
Interpretation:
The number of nitrogen‑hydrogen bonds present in primary amine has to be given.
Concept Introduction:
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the
(b)
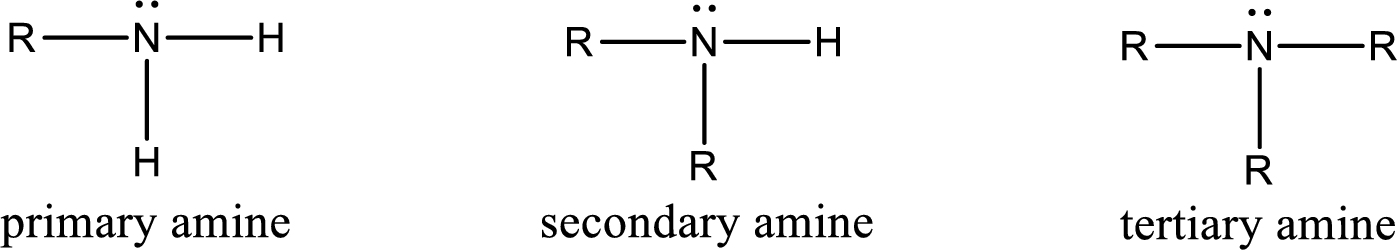
Interpretation:
The number of nitrogen‑hydrogen bonds present in secondary amine has to be given.
Concept Introduction:
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the amines are classified as primary, secondary or tertiary amine. Primary amine is the one in which only one hydrogen atom in ammonia is replaced by a hydrocarbon group. Secondary amine is the one in which only two hydrogen atoms in ammonia is replaced by a hydrocarbon group. Tertiary amine is the one in which all three hydrogen atoms in ammonia is replaced by a hydrocarbon group. The generalized structural formula for all the amines is,
(c)
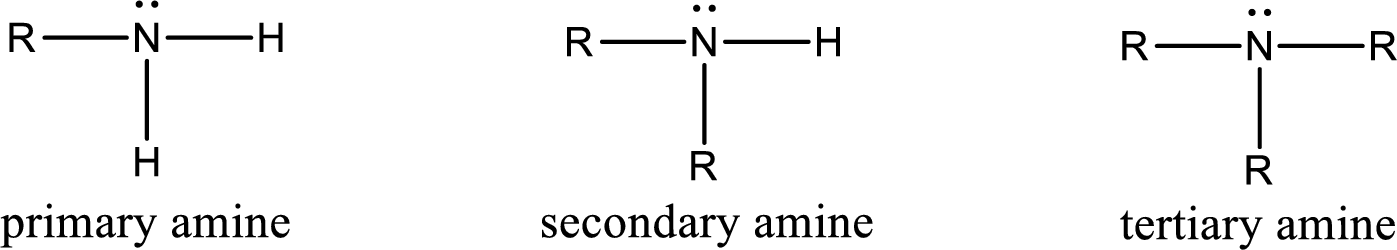
Interpretation:
The number of nitrogen‑hydrogen bonds present in tertiary amine has to be given.
Concept Introduction:
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the amines are classified as primary, secondary or tertiary amine. Primary amine is the one in which only one hydrogen atom in ammonia is replaced by a hydrocarbon group. Secondary amine is the one in which only two hydrogen atoms in ammonia is replaced by a hydrocarbon group. Tertiary amine is the one in which all three hydrogen atoms in ammonia is replaced by a hydrocarbon group. The generalized structural formula for all the amines is,
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Ninhydrin is a compound that is commonly used in forensic identification, because it turns purple in contact with the amino acids often found in sweat residue. Using the structure provided, answer the questions below. b. How many carboxylic acid functional groups are present? Explain your answer OH Œ OH 2arrow_forwardIdentify the functional groups in the following molecules. (Use names from the table below. List each class of functional group only once. If there are fewer than 3 functional groups, leave an appropriate number of answer boxes empty.) а) NH2 phenylalanine CH3 .CH3 b) ÕH ephedrinearrow_forwardDescribe any two heteropolysaccharides in terms of: a. The types of monosaccharides involved b. types of glycosidic linkages, and c. possible hydrolysatesarrow_forward
- Salol, the phenyl ester of salicylic acid, is used as an intestinal antiseptic. Draw the structure of phenyl salicylate.arrow_forwardNinhydrin is a compound that is commonly used in forensic identification, because it turns purple in contact with the amino acids often found in sweat residue. Using the structure provided, answer the questions below. b. How many carboxylic acid functional groups are present? Explain your answer. OH OHarrow_forwardClassify the following lipid (choose all that apply).CHOOSE ALL THAT APPLY a. omega-6 b. trans alkene(s) c. omega-3 d. wax ester e. saturated f. monounsaturated g. fatty acid h. triglyceride i. steroid j. polyunsaturated k. cis alkene(s)arrow_forward
- Propose a scheme to separate and isolate a mixture containing 4-aminobenzoate, 1,2,4,5-tetrachlorobenzene, and napthalene. Isolate two of the three solids by extraction. (please explain what solvents to use and the process step by step)arrow_forwardAssign a name to each of the following monosaccharides. Use D and L designations. H b. CH 2OH a. C=0 C=0 HO-C-H H-C-OH HO-C-H Н-С-Он CH,OH CH 2OH a. b. Circle all chiral centers for the monosaccharides shown. С. d. True or False: The molecules shown can rotate plane polarized light?arrow_forwardMestranol, sold under the brand names Enovid, Norinyl, and Ortho-Novum among others, is an estrogen medication which has been used in birth control pills. Select all of the functional group families, to which mestranol belongs. Choose one or more: O A. ester OB. alkene OC. ether O D. thiol O E. alcohol OF. arene OG. aldehyde OH. ketone O 1. alkyne OHarrow_forward
- Identify the component monosaccharides of each of the following compounds and describe the type of glycosidic linkage in each.arrow_forwardWhat are the functional groups that can be found in the molecule? A. Ether, secondary amine, alkene (Ether, amina sekundar, alkena) B. Ether, primary amine, alkene (Eter, amina utama, alkena) C. Ether, amide, alkene (Eter, amida, alkena) D. Ester, primary amine, alkene (Ester, amina utama, alkena)arrow_forwardWhat is the classification of the following structure? CH3 H;C CH CH-OН CH, OA Primary Alcohol O B. Secondary Alcohol OC. Tertiary Alcohol OD.Phenolarrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





