
(a)
Interpretation:
The product formed when given amide is treated with H2O and HCl should be determined:
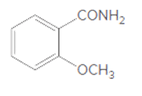
Concept Introduction:
(b)
Interpretation:
The product formed when given amide is treated with H2O and HCl should be determined:
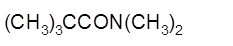
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester or amide.
Amines are the organic compounds with general chemical formula of R-NH2 or R-NH-R whereas carboxylic acids are the organic molecules with R-COOH as general chemical formula.
(c)
Interpretation:
The product formed when given amide is treated with H2O and HCl should be determined:
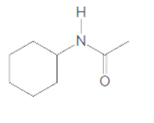
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester or amide.
Amines are the organic compounds with general chemical formula of R-NH2 or R-NH-R whereas carboxylic acids are the organic molecules with R-COOH as general chemical formula.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
General, Organic, and Biological Chemistry - 4th edition
- What ammonium salt is formed when each amine is treated with HCl? Draw the structure of the resulting salt.arrow_forwardGive an acceptable name for each amine.arrow_forwardMany drugs are sold as their hydrochloride salts (R2NH2 + Cl−), formed by reaction of an amine (R2NH) with HCl . Question: Draw the product (a hydrochloride salt) formed by reaction ofacebutolol with HCl. Acebutolol is a β blocker used to treat high bloodpressure.arrow_forward
- Draw the structure of a compound of molecular formula C4H11NO that ts each description: (a) a compound that contains a 1° amine and a 3° alcohol; (b) a compound that contains a 3° amine and a 1° alcohol.arrow_forwardamines Name and classify each compoundarrow_forwardGive each of the following amines an IUPAC name: a. b. c.arrow_forward
- Give an acceptable name for each amide.arrow_forwardSphingomyelins, a group of lipids that resemble the membrane phospholipids discussed in Section 3.7, are a major component of the myelin sheath, the insulating layer that surrounds a nerve fiber. (A) What functional groups are present in sphingomyelin X? (B) Classify any alcohol, amine, and amide as 1, 2, or 3. (C) Label the polar head and non polar tails of X.arrow_forwardFenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid (CH3CO2H)?arrow_forward
- Draw the products formed when attached carbonyl compound reacts with the following amines: [1] CH3CH2CH2NH2; [2] (CH3CH2)2NH.arrow_forwardWhich of the following is true of hydrogen bonding in amines? a) Amines do not form hydrogen bonds because there is no difference in the electronegativities of nitrogen and hydrogen. b) The N—H> N hydrogen bond is stronger than the O—H> O hydrogen bond. c) The N—H> N hydrogen bond is the same strength as that of O—H> O hydrogen bond. d) The N—H> N hydrogen bond is weaker than the O—H> O hydrogen bond.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





