
(a)
Interpretation:
The product formed when given amide is treated with H2O and NaOH should be determined:
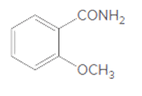
Concept Introduction:
(b)
Interpretation:
The product formed when given amide is treated with H2O and NaOH should be determined:
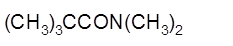
Concept Introduction:
Organic compounds are the compounds that are mainly composed C and H atoms. The branch of chemistry that deals with the preparation, reactions and properties of organic compounds. The molecular formula of an organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester or amide.
Amines are the organic compounds with general chemical formula of R-NH2 or R-NH-R, whereas, carboxylic acids are the organic molecules with R-COOH as general chemical formula.
(c)
Interpretation:
The product formed when given amide is treated with H2O and NaOH should be determined:
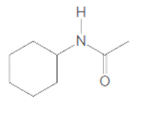
Concept Introduction:
Organic compounds are the compounds that are mainly composed C and H atoms. The branch of chemistry that deals with the preparations, reactionsand properties of organic compounds. The molecular formula of an organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester or amide.
Amines are the organic compounds with general chemical formula of R-NH2 or R-NH-R, whereas, carboxylic acids are the organic molecules with R-COOH as general chemical formula.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Give an acceptable name for each amide.arrow_forwardWhat carbonyl compound and amine are formed by the hydrolysis of each compound?arrow_forwardDraw the structure of the nitrogen-containing compound produced when each of the amides in Problem 17-140 is hydrolyzed with NaOH present (basic conditions).arrow_forward
- Draw the products formed when the given amides are treated with aqueous potassium hydroxide. Please draw the products as skeletal structures. Thanks!arrow_forwardThe –NHCOR group of an amide is an activating group, but it is not as strongly activating as NH2. (a) Explain why it is an activating group. (b) Explain why it is less activating than NH2.arrow_forwardWhat products are formed when all of the amide and ester bonds are hydrolyzed in each of the following compounds? Tamiu [part (a)] is the trade name of the antiviral agent oseltamivir, thought to be the most effective agent in treating inuenza. Aspartame [part (b)] is the articial sweetener used in Equal and many diet beverages. One of the products ofthis hydrolysis reaction is the amino acid phenylalanine. Infants afflicted with phenylketonuria cannot metabolize this amino acid, so it accumulates, causing mental retardation. When the affliction is identied early, a diet limiting the consumption of phenylalanine (and compounds like aspartame that are converted to it) can make a normal life possible.arrow_forward
- Draw the structural formula for each amides. - N-Cycloclohexylacetamide - Benzamidearrow_forwardFenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid (CH3CO2H)?arrow_forwardWhat amine is formed by reduction of each amide?arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


