
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.7, Problem 17.16P
Ibuprofen is another pain reliever that is a carboxylic acid. (a) Draw the acid—base reaction that occurs
when ibuprofen is treated with NaOH. (b) What form of ibuprofen (neutral or ionic) exists in the stomach?(c) What form of ibuprofen exists in the intestines?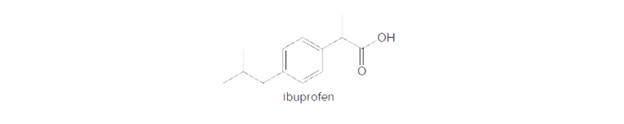
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the role of phenolphthalein in the neutralization reaction? Draw the structure of phenolphthalein under acidic and basic conditions.
Because amines are basic, they can often accept a proton. Draw the protonated structure of n-propylamine.
The hydrolysis of an amide in acidic conditions forms
A. a carboxylate salt and an alcohol
B. a carboxylate salt and an amine
C. an alcohol and an amine salt (an ammonium ion)
D. a carboxylic acid and an amine salt (an ammonium ion)
Chapter 17 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 17.1 - Draw out each compound to clearly show what groups...Ch. 17.1 - Prob. 17.2PCh. 17.1 - (a) Label each functional group in lisinopril, a...Ch. 17.2 - Give the IUPAC name for each compound. a. b. c.Ch. 17.2 - Give the structure corresponding to each IUPAC...Ch. 17.2 - Prob. 17.5PCh. 17.2 - Give an acceptable name for each ester. a. CH3(...Ch. 17.2 - Prob. 17.6PCh. 17.2 - Prob. 17.7PCh. 17.2 - Prob. 17.8P
Ch. 17.2 - Prob. 17.9PCh. 17.3 - Which compound in each pair has the higher boiling...Ch. 17.4 - Prob. 17.11PCh. 17.5 - In addition to ethyl butanoate (Section 17.5),...Ch. 17.6 - Prob. 17.3PPCh. 17.6 - Prob. 17.13PCh. 17.6 - Which compound ¡n each pair ¡s more water soluble?...Ch. 17.6 - Prob. 17.5PPCh. 17.6 - Prob. 17.14PCh. 17.6 - Prob. 17.15PCh. 17.7 - Ibuprofen is another pain reliever that is a...Ch. 17.8 - Prob. 17.6PPCh. 17.8 - Prob. 17.17PCh. 17.8 - Prob. 17.18PCh. 17.8 - Prob. 17.7PPCh. 17.8 - Prob. 17.19PCh. 17.9 - Prob. 17.8PPCh. 17.9 - Prob. 17.20PCh. 17.9 - Prob. 17.21PCh. 17.9 - Prob. 17.22PCh. 17.9 - Prob. 17.9PPCh. 17.9 - Prob. 17.23PCh. 17.9 - Prob. 17.24PCh. 17.10 - Prob. 17.10PPCh. 17.10 - Prob. 17.25PCh. 17.11 - Prob. 17.26PCh. 17 - Prob. 27PCh. 17 - Prob. 28PCh. 17 - Prob. 29PCh. 17 - Prob. 30PCh. 17 - Prob. 31PCh. 17 - Prob. 32PCh. 17 - Prob. 33PCh. 17 - Prob. 34PCh. 17 - Prob. 35PCh. 17 - Give an acceptable name for each ester. a. CH3CO2(...Ch. 17 - Prob. 37PCh. 17 - Prob. 38PCh. 17 - Prob. 39PCh. 17 - Prob. 40PCh. 17 - Prob. 41PCh. 17 - Prob. 42PCh. 17 - Prob. 43PCh. 17 - Draw the structure corresponding to each name. a....Ch. 17 - Draw the structure corresponding to each name. a....Ch. 17 - Draw the structure corresponding to each name. a....Ch. 17 - Prob. 47PCh. 17 - Prob. 48PCh. 17 - Prob. 49PCh. 17 - Prob. 50PCh. 17 - Prob. 51PCh. 17 - Prob. 52PCh. 17 - Prob. 53PCh. 17 - Prob. 54PCh. 17 - What ester is formed when butanoic acid...Ch. 17 - Prob. 56PCh. 17 - Prob. 57PCh. 17 - Prob. 58PCh. 17 - Prob. 59PCh. 17 - Prob. 60PCh. 17 - Prob. 61PCh. 17 - Prob. 62PCh. 17 - Prob. 63PCh. 17 - Prob. 64PCh. 17 - Prob. 65PCh. 17 - Prob. 66PCh. 17 - What products are formed when each ester is...Ch. 17 - Prob. 68PCh. 17 - Prob. 69PCh. 17 - Prob. 70PCh. 17 - Prob. 71PCh. 17 - Prob. 72PCh. 17 - Prob. 73PCh. 17 - Prob. 74PCh. 17 - Draw the products formed in each reaction. a. b....Ch. 17 - Draw the products formed in each reaction. a. b....Ch. 17 - Answer the following questions about A, depicted...Ch. 17 - Answer the following questions about B, depicted...Ch. 17 - Prob. 79PCh. 17 - Prob. 80PCh. 17 - Prob. 81PCh. 17 - Prob. 82PCh. 17 - Prob. 83PCh. 17 - Prob. 84PCh. 17 - Prob. 85PCh. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - Prob. 89CPCh. 17 - Lactams can be hydrolyzed with base, just like...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What are the products of the acid hydrolysis of the local anesthetic lidocaine?arrow_forward1. Differentiate volatile oil from fixed oil (table)2. Which among the crude volatile oil drugs exhibit a hallucinogenic (amphetamine-like) properly and is commonly used by prison inmates.arrow_forwardDraw the molecules involved in the synthesis of aspirin. Is the forward reaction hydrolysis or condensation? Is the reverse reaction hydrolysis or condensation? Describe what occurs un each type of reaction with respect to acetylsalicylic acid.arrow_forward
- Could I get help with this question? Thanks in advance!arrow_forwardHow does would NaOH react vs LDA in this reaction?arrow_forwardGive the IUPAC or common name of the amine formed in the reaction below between an amine salt and a base? (CH3)2NH2+Cl−+NaOH→ ?+NaCl+H2O(CH3)2NH2+Cl−+NaOH→ ?+NaCl+H2Oarrow_forward
- Draw the conjuate acid of a ketone compoundarrow_forwardUsing the data in Appendix C, determine which of the following bases is strong enough to deprotonate acetonitrile (CH3CN), so that equilibrium favors the products: (a) NaH; (b) Na2CO3; (c) NaOH; (d) NaNH2; (e) NaHCO3.arrow_forwardN-p-hydroxyphenylethanamide is commonly known as a. acetaminophen b. acetamide c. acetanilide d. formamide High molar mass amines have __________ odor. a.strong ammoniacal b.fruity c.fishy d.obnoxious Trimethyl amine has _________ odor. a.obnoxious b.fishy c. ammoniacal d. fruityarrow_forward
- a. What pH would you make the water layer to cause the carboxylic acid to dissolve in the water layer and the amine to dissolve in the ether layer? b. What pH would you make the water layer to cause the carboxylic acid to dissolve in the ether layer and the amine to dissolve in the water layer?arrow_forwardExplain why the C-3 OH group of vitamin C is more acidic than the C-2 OH group.arrow_forwardDraw the structures of both maleic and fumaric acids and explain the differences?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY