
Interpretation:
The structural feature which distinguishes
Concept introduction:
Both aldehydes and ketones are oxygen-containing functional groups.
Answer to Problem 49A
Aldehydes contain one alkyl chain attached to the carbonyl group while ketones contain two alkyl chains attached to the carbonyl group.
Explanation of Solution
Aldehydes have a functional group -CHO and ketones have −CO-. So in both, the common group is carbonyl group.
The structures are represented as follows:
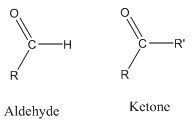
In aldehydes, the carbonyl is attached to hydrogen from one side and the other side can be hydrogen or alkyl chain. But in ketones, the carbonyl is attached to two alkyl groups.
Aldehyde functional group is always at the end of the carbon chain but the ketonic group is never attached at the end of the carbon chain.
Thus, aldehydes and ketones contain carbonyl group which makes both very much similar in reactivity and difference in alkyl groups, due to which there are some changes in physical properties.
Chapter 20 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





