
Concept explainers
(a)
Interpretation: The family of given organic compound needs to be determined.
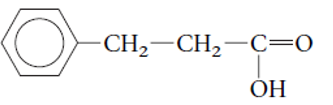
Concept Introduction:
The IUPAC rules are purposed to names different alkanes and alkyl halides.
(a)
Answer to Problem 68A
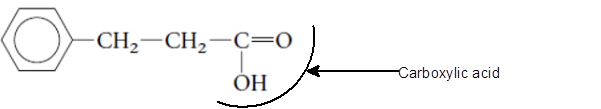
Explanation of Solution
The functional group is the atom or group of atom which is responsible for the specific chemical and physical properties in the molecule. In the given compound there is one −COOH group bonded with C atom of molecule. The −COOH group is called as carboxylic acid group.
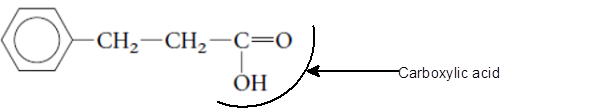
(b)
Interpretation: The family of given organic compound needs to be determined.
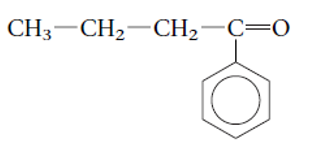
Concept Introduction: Alkanes are the saturated hydrocarbons which are mainly composed of C and H atoms. The carbon atoms are bonded in straight chain or branched chain. Alkyl halides are the halogens derivatives of alkanes. They have certain heteroatoms as Cl, I, Br or F along with C and H atoms.
The IUPAC rules are purposed to names different alkanes and alkyl halides.
(b)
Answer to Problem 68A
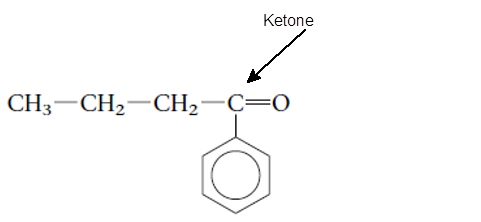
Explanation of Solution
The functional group is the atom or group of atom which is responsible for the specific chemical and physical properties in the molecule. In the given compound there is one >C=O group bonded with one alkyl and one
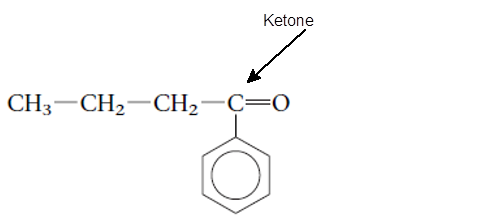
(c)
Interpretation: The family of given organic compound needs to be determined.
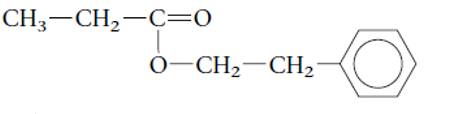
Concept Introduction: Alkanes are the saturated hydrocarbons which are mainly composed of C and H atoms. The carbon atoms are bonded in straight chain or branched chain. Alkyl halides are the halogens derivatives of alkanes. They have certain heteroatoms as Cl, I, Br or F along with C and H atoms.
The IUPAC rules are purposed to names different alkanes and alkyl halides.
(c)
Answer to Problem 68A
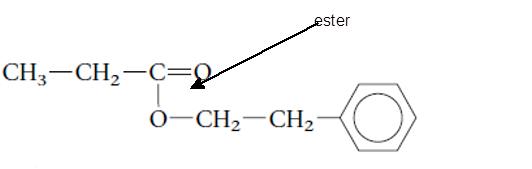
Explanation of Solution
The functional group is the atom or group of atom which is responsible for the specific chemical and physical properties in the molecule. In the given compound there is one −COO- group bonded with one alkyl and one aromatic ring. The −COO- group is called as ester group.
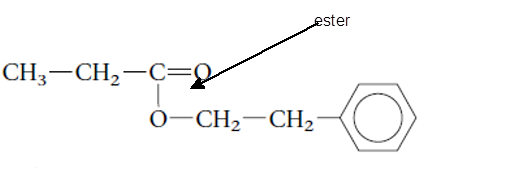
(d)
Interpretation: The family of given organic compound needs to be determined.
Concept Introduction: Alkanes are the saturated hydrocarbons which are mainly composed of C and H atoms. The carbon atoms are bonded in straight chain or branched chain. Alkyl halides are the halogens derivatives of alkanes. They have certain heteroatoms as Cl, I, Br or F along with C and H atoms.
The IUPAC rules are purposed to names different alkanes and alkyl halides.
(d)
Answer to Problem 68A
Explanation of Solution
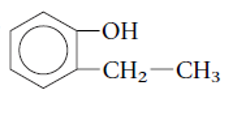
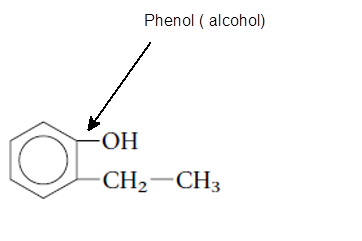
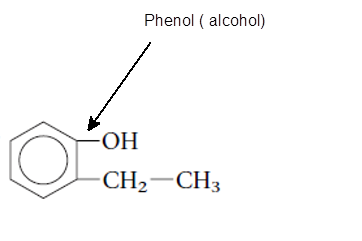
The functional group is the atom or group of atom which is responsible for the specific chemical and physical properties in the molecule. In the given compound there is one −OH group bonded with one aromatic ring. The −OH group is called as alcoholic group and if −OH group is bonded on aromatic ring it is called as phenolic group.
Chapter 20 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





